A. ATOMIC STRUCTURE
The atom is the smallest particle of an element that takes part in a chemical reaction. The atom is made up of three subatomic particles:
- Protons
- Electrons
- Neutrons
(i) Protons
- The proton is positively charged.
- It is found in the centre of an atom called the nucleus.
- It has a relative mass of 1.
- The number of protons in an atom of an element is its Atomic number.
(ii) Electrons
- The electron is negatively charged.
- It is found in fixed regions surrounding the centre of an atom called energy levels or orbitals.
- It has a relative mass of 1/1840.
- The number of protons and electrons in an atom of an element is always equal.
(iii) Neutrons
- The neutron is neither positively nor negatively charged; it is neutral.
- Like protons, it is found in the centre of an atom called the nucleus.
- It has a relative mass of 1.
- The number of protons and neutrons in an atom of an element is its Mass number.
Diagram showing the relative positions of protons, electrons and neutrons in an atom of an element
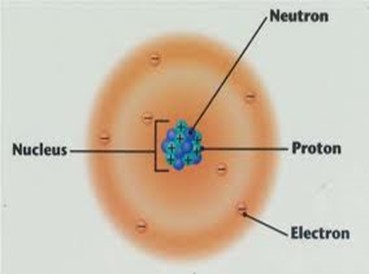
Diagram showing the relative positions of protons, electrons and neutrons in an atom of Carbon

The table below shows the atomic structure of the first twenty elements.
| Element | Symbol | Protons | Electrons | Neutrons | Atomic number | Mass number |
|---|---|---|---|---|---|---|
| Hydrogen | H | 1 | 1 | 0 | 1 | 1 |
| Helium | He | 2 | 2 | 2 | 2 | 4 |
| Lithium | Li | 3 | 3 | 4 | 3 | 7 |
| Beryllium | Be | 4 | 4 | 5 | 4 | 9 |
| Boron | B | 5 | 5 | 6 | 5 | 11 |
| Carbon | C | 6 | 6 | 6 | 6 | 12 |
| Nitrogen | N | 7 | 7 | 7 | 7 | 14 |
| Oxygen | O | 8 | 8 | 8 | 8 | 16 |
| Fluorine | F | 9 | 9 | 10 | 9 | 19 |
| Neon | Ne | 10 | 10 | 10 | 10 | 20 |
| Sodium | Na | 11 | 11 | 12 | 11 | 23 |
| Magnesium | Mg | 12 | 12 | 12 | 12 | 24 |
| Aluminium | Al | 13 | 13 | 14 | 13 | 27 |
| Silicon | Si | 14 | 14 | 14 | 14 | 28 |
| Phosphorus | P | 15 | 15 | 16 | 15 | 31 |
| Sulphur | S | 16 | 16 | 16 | 16 | 32 |
| Chlorine | Cl | 17 | 17 | 18 | 17 | 35 |
| Argon | Ar | 18 | 18 | 22 | 18 | 40 |
| Potassium | K | 19 | 19 | 20 | 19 | 39 |
| Calcium | Ca | 20 | 20 | 20 | 20 | 40 |
Most atoms of elements exist as isotopes.
Isotopes are atoms of the same element, having the same number of protons/atomic number but different number of neutrons/mass number.
By convention, isotopes are written with the mass number as superscript and the atomic number as subscript to the left of the chemical symbol of the element, i.e.
mass number mn X (symbol of element)
Below is the conventional method of writing the first twenty elements showing the mass numbers and atomic numbers:
11H 42He 73Li 94Be 115B 126C
147N 168O 199F 2010Ne 2311Na 2412Mg
2713Al 2814Si 3115P 3216S 3517Cl 4018Ar
3919K 4020Ca
The table below shows some common natural isotopes of some elements.
| Element | Isotopes | Protons | Electrons | Neutrons | Atomic number | Mass number |
|---|---|---|---|---|---|---|
| Hydrogen | 11H 21H (deuterium) 31H (tritium) | 1 1 1 | 1 1 1 | 0 2 3 | 1 1 1 | 1 2 3 |
| Chlorine | 3517Cl 3717Cl | 17 17 | 17 17 | 18 20 | 17 17 | 35 37 |
| Potassium | 3919K 4019K 4119K | 19 19 19 | 19 19 19 | 20 21 22 | 19 19 19 | 39 40 41 |
| Oxygen | 168O 188O | 8 8 | 8 8 | 8 10 | 8 8 | 16 18 |
| Uranium | 23592U 23892U | 92 92 | 92 92 | 143 146 | 92 92 | 235 238 |
| Neon | 2210Ne 2010Ne 2110Ne | 10 10 10 | 10 10 10 | 12 10 11 | 10 10 10 | 22 20 21 |
The mass of an average atom is very small (10-22 g). Masses of atoms are therefore expressed in relation to a chosen element.
The atom recommended is the 12C isotope whose mass is arbitrarily assigned as 12.000 atomic mass units (a.m.u).
All other atoms are compared to the mass of the 12C isotope to give the relative atomic mass. The relative atomic mass (RAM) is therefore defined as “the mass of an average atom of an element compared to 1/12 of an atom of the 12C isotope whose mass is arbitrarily fixed as 12.000 atomic mass units (a.m.u)“, i.e.:
RAM = mass of atom of an element / 1/12 of one atom of 12C isotope
Accurate relative atomic masses (RAM) are obtained from the mass spectrometer. A mass spectrometer determines the isotopes of the element and their relative abundance/availability.
Using the relative abundances/availability of the isotopes, the relative atomic mass (RAM) can be determined/calculated as in the examples below.
Chlorine occurs as 75% 3517Cl and 25% 3717Cl isotopes. Calculate the relative atomic mass of Chlorine.
Working
100 atoms of chlorine contain 75 atoms of 3517Cl isotopes
100 atoms of chlorine contain 25 atoms of 3717Cl isotopes
Therefore;
RAM of chlorine = (75/100 × 35) + (25/100 × 37) = 35.5
Note that:
Relative atomic mass has no units.
More atoms of chlorine exist as 3517Cl (75%) than as 3717Cl (25%), therefore RAM is nearer to the more abundant isotope.
Calculate the relative atomic mass of potassium given that it exists as:
93.1% 3919K, 0.01% 4019K, 6.89% 4119K
Working
100 atoms of potassium contain 93.1 atoms of 3919K isotopes
100 atoms of potassium contain 0.01 atoms of 4019K isotopes
100 atoms of potassium contain 6.89 atoms of 4119K isotopes
Therefore;
RAM of potassium = (93.1/100 × 39) + (0.01/100 × 40) + (6.89/100 × 41)
Note that:
Relative atomic mass has no units.
More atoms of potassium exist as 3919K (93.1%), therefore RAM is nearer to the more abundant 3919K isotope.
Calculate the relative atomic mass of Neon given that it exists as:
90.92% 2010Ne, 0.26% 2110Ne, 8.82% 2210Ne
Working
100 atoms of Neon contain 90.92 atoms of 2010Ne isotopes
100 atoms of Neon contain 0.26 atoms of 2110Ne isotopes
100 atoms of Neon contain 8.82 atoms of 2210Ne isotopes
Therefore;
RAM of Neon = (90.92/100 × 20) + (0.26/100 × 21) + (8.82/100 × 22)
Note that:
Relative atomic mass has no units.
More atoms of Neon exist as 2010Ne (90.92%), therefore RAM is nearer to the more abundant 2010Ne isotope.
Calculate the relative atomic mass of Argon given that it exists as:
90.92% 2010Ne, 0.26% 2110Ne, 8.82% 2210Ne
NB
The relative atomic mass is a measure of the masses of atoms. The higher the relative atomic mass, the heavier the atom.
Electrons are found in energy levels/orbitals.
An energy level is a fixed region around/surrounding the nucleus of an atom occupied by electrons of the same (potential) energy.
By convention, energy levels are named 1, 2, 3… outwards from the region nearest to the nucleus.
Each energy level is occupied by a fixed number of electrons:
- The 1st energy level is occupied by a maximum of two electrons.
- The 2nd energy level is occupied by a maximum of eight electrons.
- The 3rd energy level is occupied by a maximum of eight electrons (or eighteen electrons if available).
- The 4th energy level is occupied by a maximum of eight electrons (or eighteen or thirty-two electrons if available).
This arrangement of electrons in an atom is called electron configuration or structure.
By convention, the electron configuration/structure of an atom of an element can be shown in the form of a diagram using either cross (x) or dot (●) to represent electrons.
Practice examples drawing electronic configurations
a) 11H has – in nucleus 1 proton and 0 neutrons
– 1 electron in the 1st energy level thus:
Nucleus
Energy levels
Electrons (represented by cross (x))
Electronic structure of Hydrogen is thus: 1:
b) 42He has – in nucleus 2 protons and 2 neutrons
– 2 electrons in the 1st energy level thus:
Nucleus
Energy levels
Electrons (represented by cross (x))
Electronic structure of Helium is thus: 2:
c) 73Li has – in nucleus 3 protons and 4 neutrons
– 2 electrons in the 1st energy level
– 1 electron in the 2nd energy level thus
Nucleus
Energy levels
Electrons (represented by cross (x))
Electronic structure of Lithium is thus: 2:1
d) 94Be has – in nucleus 4 protons and 5 neutrons
– 2 electrons in the 1st energy level
– 2 electrons in the 2nd energy level thus
Nucleus
Energy levels
Electrons (represented by cross (x))
Electronic structure of Beryllium is thus: 2:2
e) 115B has – in nucleus 5 protons and 6 neutrons
– 2 electrons in the 1st energy level
– 3 electrons in the 2nd energy level thus
Nucleus
Energy levels
Electrons (represented by cross (x))
Electronic structure of Boron is thus: 2:3
f) 126C has – in nucleus 6 protons and 6 neutrons
– 2 electrons in the 1st energy level
– 4 electrons in the 2nd energy level thus
Nucleus
Energy levels
Electrons (represented by cross (x))
Electronic structure of Carbon is thus: 2:4
g) 147N has – in nucleus 7 protons and 7 neutrons
– 2 electrons in the 1st energy level
– 5 electrons in the 2nd energy level thus
Nucleus
Energy levels
Electrons (represented by cross (x))
Electronic structure of Nitrogen is thus: 2:5
h) 168O has – in nucleus 8 protons and 8 neutrons
– 2 electrons in the 1st energy level
– 6 electrons in the 2nd energy level thus
Nucleus
Energy levels
Electrons (represented by cross (x))
Electronic structure of Oxygen is thus: 2:6
i) 199F has – in nucleus 9 protons and 10 neutrons
– 2 electrons in the 1st energy level
– 7 electrons in the 2nd energy level thus
Nucleus
Energy levels
Electrons (represented by cross (x))
Electronic structure of Fluorine is thus: 2:7
j) 2010Ne has – in nucleus 10 protons and 10 neutrons
– 2 electrons in the 1st energy level
– 8 electrons in the 2nd energy level thus
Nucleus
Energy levels
Electrons (represented by cross (x))
Electronic structure of Neon is thus: 2:8
k) 2311Na has – in nucleus 11 protons and 12 neutrons
– 2 electrons in the 1st energy level
– 8 electrons in the 2nd energy level
– 1 electron in the 3rd energy level thus
Nucleus
Energy levels
Electrons (represented by dot (●))
Electronic structure of Sodium is thus: 2:8:1
l) 2412Mg has – in nucleus 12 protons and 12 neutrons
– 2 electrons in the 1st energy level
– 8 electrons in the 2nd energy level
– 2 electrons in the 3rd energy level thus
Nucleus
Energy levels
Electrons (represented by dot (●))
Electronic structure of Magnesium is thus: 2:8:2
m) 2713Al has – in nucleus 13 protons and 14 neutrons
– 2 electrons in the 1st energy level
– 8 electrons in the 2nd energy level
– 3 electrons in the 3rd energy level thus
Nucleus
Energy levels
Electrons (represented by dot (●))
Electronic structure of Aluminium is thus: 2:8:3
n) 2814Si has – in nucleus 14 protons and 14 neutrons
– 2 electrons in the 1st energy level
– 8 electrons in the 2nd energy level
– 4 electrons in the 3rd energy level thus
Nucleus
Energy levels
Electrons (represented by dot (●))
Electronic structure of Silicon is thus: 2:8:4
o) 3115P has – in nucleus 15 protons and 16 neutrons
– 2 electrons in the 1st energy level
– 8 electrons in the 2nd energy level
– 5 electrons in the 3rd energy level thus
Nucleus
Energy levels
Electrons (represented by dot (●))
Electronic structure of Phosphorus is thus: 2:8:5
p) 3216S has – in nucleus 16 protons and 16 neutrons
– 2 electrons in the 1st energy level
– 8 electrons in the 2nd energy level
– 6 electrons in the 3rd energy level thus
Nucleus
Energy levels
Electrons (represented by dot (●))
Electronic structure of Sulphur is thus: 2:8:6
q) 3517Cl has – in nucleus 17 protons and 18 neutrons
– 2 electrons in the 1st energy level
– 8 electrons in the 2nd energy level
– 7 electrons in the 3rd energy level thus
Nucleus
Energy levels
Electrons (represented by dot (●))
Electronic structure of Chlorine is thus: 2:8:7
r) 4018Ar has – in nucleus 18 protons and 22 neutrons
– 2 electrons in the 1st energy level
– 8 electrons in the 2nd energy level
– 8 electrons in the 3rd energy level
Electronic structure of Argon is thus: 2:8:8
s) 3919K has – in nucleus 19 protons and 20 neutrons
– 2 electrons in the 1st energy level
– 8 electrons in the 2nd energy level
– 8 electrons in the 3rd energy level
– 1 electron in the 4th energy level thus
Nucleus
Energy levels
Electrons (represented by dot (●))
Electronic structure of Potassium is thus: 2:8:8:1
t) 4020Ca has – in nucleus 20 protons and 20 neutrons
– 2 electrons in the 1st energy level
– 8 electrons in the 2nd energy level
– 8 electrons in the 3rd energy level
– 2 electrons in the 4th energy level thus
Nucleus
Energy levels
Electrons (represented by dot (●))
Electronic structure of Calcium is thus: 2:8:8:2





9 Comments