1.0
Introduction
2.0
Objective
- Main Content
- Theory
- Procedure
4.0 Conclusion
5.0 Summary
6.0 Tutor Marked Assignments
1.0
Introduction
Most chemical reactions require two or more reactants. Typically, one of the reactants is used up before the other, at which time the reaction stops. The chemical that is used up is called the limiting reactant while the other reactant is present in excess. If both reactants are present in exactly the right amount to react completely, without either in excess, the amounts of reactants are said to be in a stoichiometric ratio to each other. The stoichiometric ratio is the mole ratio of the reactants, or reactants to products, as determined by the coefficients in the balanced chemical equation. Since the limiting reactant will determine the amount of product that can be produced during a reaction, it is important to be able to calculate which reactant is the limiting reactant. There are several ways to do this, but each starts with a balanced chemical equation so that the stoichiometry of the reaction is known.
2.0
Objectives
At the end of this unit you will achieve the following:
- Explain limiting reaction
- Explain stoichiometric ratio
- Percent yield
- Perform an experiment to determine limiting reactant using percent yield
- Main Content
- Theory
Many times an analysis will be based on the fact that you have a limiting reactant involved in the chemical reaction. The limiting reactant determines the percent yield of product. Because chemicals react stoichiometrically, only a limited amount of product forms from given amounts of reactants. For example, the analysis of a sample containing soluble sulfate salt such as sodium sulfate can be performed by dissolving the sample in water and adding a
solution containing barium chloride to form an insoluble barium sulfate salt and sodium chloride which is soluble.

Since all of the salts, except , are soluble, the net ionic equation for the chemical reaction is
, are soluble, the net ionic equation for the chemical reaction is 
.
One mole of barium ion, from 
(244.2g) in solution, reacts with 1 mole of sulfate ion, from 1 mole of  (142.lg) in solution, to produce 1 mole of barium sulfate precipitate (233.4g) if the reaction takes place completely. The reaction is referred to as being quantitative since it will allow the determination of the amount of sodium sulfate in the sample. Sodium sulfate is the limiting reactant and to ensure that a total, complete reaction takes place an excess of barium chloride solution is added.
(142.lg) in solution, to produce 1 mole of barium sulfate precipitate (233.4g) if the reaction takes place completely. The reaction is referred to as being quantitative since it will allow the determination of the amount of sodium sulfate in the sample. Sodium sulfate is the limiting reactant and to ensure that a total, complete reaction takes place an excess of barium chloride solution is added.
A 1.000 g sample of unknown salt mixture containing sodium sulfate is weighed to the nearest 1mg. The number of moles of sodium sulfate are calculated as if this were a pure sample of the sodium sulfate,
1.000g 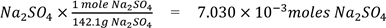
thus representing maximum number of moles of sodium sulfate which may be present in the sample. Using the balanced equation:
 ,
,
Or
=
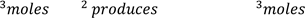
And

Thus, 1.641g represents the maximum amount of precipitate that can be formed if the sample is 100% pure. This is referred to as the theoretical yield of the product. Since the sample is not pure sodium sulfate, you will obtain less than the theoretical amount which we will call the experimental value. The percentage of sodium sulfate in the unknown can be calculated using the percent yield formula which is defined as:
represents the maximum amount of precipitate that can be formed if the sample is 100% pure. This is referred to as the theoretical yield of the product. Since the sample is not pure sodium sulfate, you will obtain less than the theoretical amount which we will call the experimental value. The percentage of sodium sulfate in the unknown can be calculated using the percent yield formula which is defined as:


Many industrial products such as food ingredients, deodorants, mineral waters, and cosmetics would use this procedure for analysis of sulfate ion.
In this experiment an unknown salt mixture containing  is analyzed for the percentage of
is analyzed for the percentage of  by adding excess
by adding excess solution to ensure completeness of reaction. The other ingredient in the mixture is a nonreactive, soluble substance which will not interfere with the quantitative reaction. The precipitate,
solution to ensure completeness of reaction. The other ingredient in the mixture is a nonreactive, soluble substance which will not interfere with the quantitative reaction. The precipitate, , will be vacuum filtered, dried, and weighed to obtain experimental yield of
, will be vacuum filtered, dried, and weighed to obtain experimental yield of  . The theoretical yield will be based on 100% activity of the salt mixture sample weighed.
. The theoretical yield will be based on 100% activity of the salt mixture sample weighed.
3.2 Procedure
Weigh a clean, empty 400 ml beaker to the nearest 0.001 g. Add about 1.0 g of the unknown salt mixture to the beaker and reweigh the beaker to the nearest 0.001 g.
Calculate the number of moles of sodium sulfate that theoretically are contained in this sample weight. Also calculate the theoretical yield of  as demonstrated in the theory section of this experiment.
as demonstrated in the theory section of this experiment.
Add 200 ml of distilled water to the beaker and stir with a stirring rod. In the hood, measure 1ml of concentrated HCl in a small graduated cylinder and with the aid of a stirring rod. Add to the solution in the beaker.
CAUTION: If spilled concentrated HC1, hydrochloric acid, is a severe skin irritant-flush affected area with large amounts of water.
In graduated cylinder, measure 50 ml of  0.5 M and add to the solution in the beaker and stir for several minutes with a stirring rod. Allow the precipitate to settle. Verify that the amount of barium chloride added to the solution does, in fact, represent an excess of the reactant. With the stirring rod in the beaker, cover the beaker with a watch glass and place the entire apparatus on a hot plate. Heat at low setting to maintain solution temperature between 80-900C
0.5 M and add to the solution in the beaker and stir for several minutes with a stirring rod. Allow the precipitate to settle. Verify that the amount of barium chloride added to the solution does, in fact, represent an excess of the reactant. With the stirring rod in the beaker, cover the beaker with a watch glass and place the entire apparatus on a hot plate. Heat at low setting to maintain solution temperature between 80-900C
for 1 hour. Check the temperature of the solution with your thermometer occasionally.
Avoid boiling the solution. Heat approximately 100ml
of distilled water in a beaker to be used as the washing solvent.
While the solution is heating, assemble the vacuum filtration apparatus. Two types of filters will be used: a Buchner funnel with a paper filter and a fritted glass filtering crucible.
The advantage of vacuum filtration is the speed of isolation for a solid product from solution. One trial will be done recovering the precipitate on the paper and a second trial will recover the solid in the filtering crucible. It will require the recorded weight of both, paper and crucible. The order in which these two isolation methods are done is not a significant factor.
Before the filtration process is started, tip the beaker and allow the precipitate to settle under the lip of the beaker. While the solution is still hot, using beaker tongs, decant the supernatant liquid trying to minimize solid transfer to the filter. Wash the precipitate several times with warm distilled water, using a rubber policeman to remove any solid adhering to the sides of the beaker. Using the wash bottle, transfer the precipitate to the filtering medium and rinse several times with the warm distilled water. Allow air. to be drawn through the filtering medium for several minutes after the last rinse. Break the vacuum seal at the filtering flask and remove the filtering medium to a clean, dry, preweighed, marked watch glass. Place the watch glass and filtering medium in drying oven for several hours or overnight. When the precipitate is dry, allow it to cool and weigh watch glass and filtering medium.
Calculate the weight of  isolated. Calculate the percent yield of the reaction. Report the average percentage yield on two trials.
isolated. Calculate the percent yield of the reaction. Report the average percentage yield on two trials.
4.0
Conclusion A limiting reactant
is the reagent that is completely consumed during a chemical reaction. Once this reagent is consumed the reaction stops. An excess reagent
is the reactant that is left over once the limiting reagent is consumed. The maximum theoretical yield of a chemical reaction is dependent upon the limiting reagent thus the one that produces the least amount of product is the limiting reagent.
5.0
Summary In this unit, you have studied about limiting reactant, excess reactant(reagent) stoichiometric ratio and percent yield .You have also performed an experiment to determine limiting reactant using percent yield.
6.0 Tutor Marked Experiment
1. Using a suitable reference source, define the following words:
- Limiting reactant
- Excess reactant
- stoichiometric ratio
2 Take the reaction: NH3 + O2
 NO + H2O. In an experiment, 3.25 g of NH3 are allowed to react with 3.50 g of O2. a. Which reactant is the limiting reagent? O2
NO + H2O. In an experiment, 3.25 g of NH3 are allowed to react with 3.50 g of O2. a. Which reactant is the limiting reagent? O2
- How many grams of NO are formed? 2.63 g NO
- How much of the excess reactant remains after the reaction? 1.76 g NH3 left
3.Consider the reaction of C6H6 + Br2
 C6H5Br + HBr
C6H5Br + HBr
- What is the theoretical yield of C6H5Br if 42.1 g of C6H6 react with 73.0 g of Br2? 71.8 g C6H5Br
- If the actual yield of C6H5Br is 63.6 g, what is the percent yield? 88.6%
4. Use the following reaction: C4H9OH + NaBr + H2SO4
 C4H9Br + NaHSO4 + H2O If 15.0 g of C4H9OH react with 22.4 g of NaBr and 32.7 g of H2SO4 to yield 17.1 g of C4H9Br, what is the percent yield of this reaction?
C4H9Br + NaHSO4 + H2O If 15.0 g of C4H9OH react with 22.4 g of NaBr and 32.7 g of H2SO4 to yield 17.1 g of C4H9Br, what is the percent yield of this reaction?
61.8%





