THERMOCHEMISTRY
1. Introduction to Energy Changes
Energy is the capacity to do work. There are various forms of energy such as heat, electric, mechanical, and chemical energy. Energy exists mainly in two types:
- Kinetic Energy (KE): the energy of motion.
- Potential Energy (PE): the stored or internal energy.
Energy, like matter, is neither created nor destroyed but can be transformed or changed from one form to another; this is known as the principle of conservation of energy. For example, electrical energy is converted into heat energy through a filament in a bulb.
Chemical and physical processes occur with either absorption or evolution (production) of energy, mainly in the form of heat.
The study of energy changes that accompany physical or chemical reactions or changes is called Thermochemistry. Physical or chemical reactions or changes that involve energy changes are called thermochemical reactions. The SI unit of energy is the Joule (J). Larger units such as KiloJoules (kJ) and MegaJoules (MJ) are also commonly used. The Joule (J) is defined as:
- The quantity of energy transferred when a force of one newton acts through a distance of one metre.
- The quantity of energy transferred when one coulomb of electric charge passes through a potential difference of one volt.
All thermochemical reactions should be carried out under standard conditions of:
- 298 K / 25°C temperature
- 101300 Pa / 101300 N/m2 / 760 mmHg / 1 atmosphere pressure
2. Exothermic and Endothermic Processes/Reactions
Some reactions or processes take place with the evolution or production of energy. These are called exothermic reactions. Others take place with the absorption of energy and are called endothermic reactions.
Practically, exothermic reactions cause a rise in temperature, which can be observed by an increase in thermometer reading or mercury/alcohol level.
Conversely, endothermic reactions cause a fall in temperature, observed by a decrease in thermometer reading or mercury/alcohol level.
To Demonstrate/Illustrate Exothermic and Endothermic Processes/Reactions
Dissolving Potassium nitrate (V) / Ammonium chloride crystals
Procedure:
Measure 20 cm3 of water in a beaker. Determine and record its initial temperature T1. Add about 1.0 g of Potassium nitrate (V) crystals into the beaker. Stir the mixture carefully and note the highest temperature reached T2. Repeat the procedure using ammonium chloride crystals instead of potassium nitrate.
Sample Results
Temperature (°C) Using Potassium nitrate (V) crystals Using Ammonium chloride crystals T2 (Final temperature) 21.0 23.0 T1 (Initial temperature) 25.0 26.0 Change in temperature (T2 – T1) -4.0 -3.0 Note:
- The initial temperature (T1) of dissolution for both potassium nitrate (V) and ammonium chloride crystals is higher than the final temperature (T2).
- The change in temperature (T2 – T1) is negative, indicating a temperature fall.
- Dissolution of both potassium nitrate (V) and ammonium chloride crystals is an endothermic process because the temperature falls, indicating absorption of heat from the surroundings.
Dissolving concentrated sulphuric (VI) acid / sodium hydroxide crystals
Procedure:
Measure 20 cm3 of water in a beaker. Determine and record its initial temperature T1. Carefully add about 1.0 g (or four pellets) of sodium hydroxide crystals into the beaker. Stir the mixture carefully and note the highest temperature reached T2. Repeat the procedure using 2 cm3 of concentrated sulphuric (VI) acid instead of sodium hydroxide crystals.
CAUTION:
- Sodium hydroxide crystals are caustic and cause painful blisters on contact with skin.
- Concentrated sulphuric (VI) acid is corrosive and causes painful wounds on contact with skin.
Sample Results
Temperature (°C) Using Sodium hydroxide pellets Using Concentrated sulphuric (VI) acid T2 (Final temperature) 30.0 32.0 T1 (Initial temperature) 24.0 25.0 Change in temperature (T2 – T1) 6.0 7.0 Note:
- The initial temperature (T1) of dissolution for both concentrated sulphuric (VI) acid and sodium hydroxide pellets is lower than the final temperature (T2).
- Dissolution of both sodium hydroxide pellets and concentrated sulphuric (VI) acid is an exothermic process because the temperature rises, indicating heat loss to the surroundings.
The above reactions demonstrate heat loss to and heat gain from the surroundings, as illustrated by the rise and fall in temperature or thermometer readings.
Dissolving potassium nitrate (V) and ammonium chloride crystals causes heat gain from the surroundings, resulting in a fall in temperature.
Dissolving sodium hydroxide pellets and concentrated sulphuric (VI) acid causes heat loss to the surroundings, resulting in a rise in temperature.
At the same temperature and pressure, heat absorbed or released is called enthalpy or heat content, denoted by H.
Energy change is measured from the heat content or enthalpy of the final and initial products. It is denoted ∆H (delta H), i.e.,
Enthalpy or energy change in heat content ∆H = Hfinal – Hinitial
For chemical reactions:
∆H = Hproducts – Hreactants
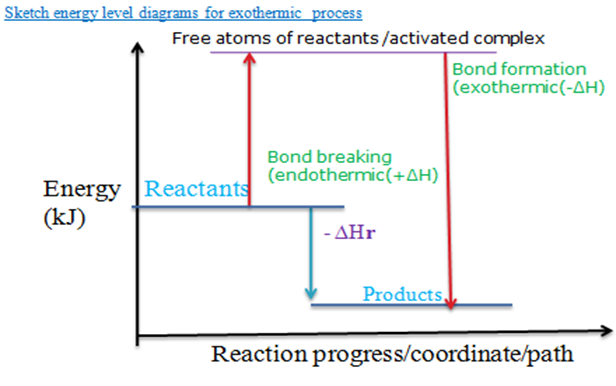
For exothermic reactions, the heat content of the reactants is higher than that of the products, so ∆H is negative (-∆H).
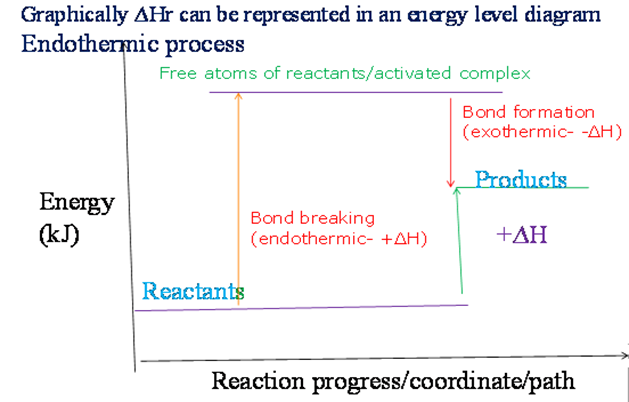
For endothermic reactions, the heat content of the reactants is lower than that of the products, so ∆H is positive (+∆H).
Graphically, in a sketch energy level diagram:
- For endothermic reactions, the heat content of the reactants is slightly lower than that of the products.
- For exothermic reactions, the heat content of the reactants is slightly higher than that of the products.
Sketch Energy Level Diagrams for Endothermic Dissolution
Energy (kJ)
H2 KNO3(aq)
+∆H = H2 – H1
H1 KNO3(s)
Reaction path/coordinate/progress
Energy (kJ)
H2 NH4Cl (aq)
+∆H = H2 – H1
H1 NH4Cl (s)
Reaction path/coordinate/progress
Sketch Energy Level Diagrams for Exothermic Dissolution
H2 NaOH (s)
Energy (kJ)
-∆H = H2 – H1
H1 NaOH (aq)
Reaction path/coordinate/progress
H2 H2SO4 (l)
Energy (kJ)
-∆H = H2 – H1
H1 H2SO4 (aq)
Reaction path/coordinate/progress
3. Energy Changes in Physical Processes
Melting/freezing/fusion/solidification and boiling/vaporization/evaporation are two important physical processes. The melting/freezing point of pure substances is fixed or constant. The boiling point of a pure substance depends on the external atmospheric pressure.
Melting or fusion is the physical change of a solid to a liquid. Freezing is the physical change of a liquid to a solid.
Melting/freezing/fusion/solidification are therefore two opposite but reversible physical processes, i.e.,
A (s) ======== A (l)
Boiling, vaporization, or evaporation is the physical change of a liquid to gas/vapour. Condensation or liquefaction is the physical change of gas/vapour to liquid. Boiling/vaporization/evaporation and condensation/liquefaction are therefore two opposite but reversible physical processes, i.e.,
B (l) ======== B (g)
Practically:
- Melting/liquefaction/fusion involves heating a solid to weaken the strong bonds holding the solid particles together. Solids consist of particles held very close by strong bonds (Kinetic Theory of Matter). On heating, these particles gain energy or heat from the surroundings to form a liquid with weaker bonds, allowing some degree of freedom. Melting/freezing/fusion is an endothermic (+∆H) process that requires or absorbs energy from the surroundings.
- Freezing/fusion/solidification involves cooling a liquid to reform or strengthen the strong bonds holding the particles close together as a solid, thus losing their degree of freedom. Freezing/fusion/solidification is an exothermic (-∆H) process that releases energy to the surroundings.
- Boiling/vaporization/evaporation involves heating a liquid to completely break/free the bonds holding the liquid particles together. Gaseous particles have a high degree of freedom. Boiling/vaporization/evaporation is an endothermic (+∆H) process that absorbs energy from the surroundings.
- Condensation/liquefaction is the reverse process of boiling/vaporization/evaporation. It involves gaseous particles losing energy to the surroundings to form a liquid. It is an exothermic (-∆H) process.
The quantity of energy required to change one mole of a solid to liquid or to form one mole of a solid from liquid at constant temperature is called the molar enthalpy/latent heat of fusion. For example:
H2O (s) → H2O (l) ∆H = +6.0 kJ mole-1 (endothermic process)
H2O (l) → H2O (s) ∆H = -6.0 kJ mole-1 (exothermic process)
The quantity of energy required to change one mole of a liquid to gas/vapour or to form one mole of a liquid from gas/vapour at constant temperature is called the molar enthalpy/latent heat of vaporization. For example:
H2O (l) → H2O (g) ∆H = +44.0 kJ mole-1 (endothermic process)
H2O (g) → H2O (l) ∆H = -44.0 kJ mole-1 (exothermic process)
Experiments Illustrating Practical Determination of Melting and Boiling Points
To determine the boiling point of water
Procedure:
Measure 20 cm3 of tap water into a 50 cm3 glass beaker. Determine and record its initial temperature. Heat the water on a strong Bunsen burner flame and record its temperature every thirty seconds for four minutes.
Sample Results
Time (seconds) 0 30 60 90 120 150 180 210 240 Temperature (°C) 25.0 45.0 85.0 95.0 96.0 96.0 96.0 97.0 98.0 Questions
1. Plot a graph of temperature against time (y-axis).
Sketch a graph of temperature against time.
Boiling point: 96°C
Temperature (°C): 25°C
Time (seconds)
2. From the graph, show and determine the boiling point of water.
Note: Water boils at 100°C at sea level/one atmosphere pressure/101300 Pa but boils below 100°C at higher altitudes. The sample results above are from Kiriari Girls High School, Embu County on the slopes of Mt Kenya in Kenya. Water here boils at 96°C.
3. Calculate the molar heat of vaporization of water (H=1.0, O=16.0).
Working:
Mass of water = density × volume = (20 × 1) / 1000 = 0.02 kg
Quantity of heat produced = mass of water × specific heat capacity × temperature change
= 0.02 kg × 4.2 kJ/kg·K × (96 – 25) = 5.964 kJ
Heat of vaporization of one mole H2O = Quantity of heat / Molar mass of H2O
= 5.964 kJ / 18 = 0.3313 kJ mole-1
To determine the melting point of candle wax
Procedure:
Weigh exactly 5.0 g of candle wax into a boiling tube. Heat it on a strong Bunsen burner flame until it completely melts. Insert a thermometer and remove the boiling tube from the flame. Stir continuously. Determine and record the temperature every 30 seconds for four minutes.
Sample Results
Time (seconds) 0 30 60 90 120 150 180 210 240 Temperature (°C) 93.0 85.0 78.0 70.0 69.0 69.0 69.0 67.0 65.0 Questions
1. Plot a graph of temperature against time (y-axis).
Sketch a graph of temperature against time.
Melting point: 69°C
Temperature (°C): 93°C
Time (seconds)
2. From the graph, show and determine the melting point of the candle wax.
4. Energy Changes in Chemical Processes
Thermochemical reactions measured at standard conditions of 298 K (25°C) and 101300 Pa / 101300 N/m2 / 1 atmosphere / 760 mmHg / 76 cmHg produce standard enthalpies denoted ∆H°.
Thermochemical reactions are named according to the type of reaction producing the energy change. Below are some thermochemical reactions:
- Standard enthalpy/heat of reaction ∆H°r
- Standard enthalpy/heat of combustion ∆H°c
- Standard enthalpy/heat of displacement ∆H°d
- Standard enthalpy/heat of neutralization ∆H°n
- Standard enthalpy/heat of solution/dissolution ∆H°s
- Standard enthalpy/heat of formation ∆H°f
(a) Standard Enthalpy/Heat of Reaction ∆H°r
The molar standard enthalpy or heat of reaction is defined as the energy or heat change when one mole of products is formed at standard conditions.
A chemical reaction involves reactants forming products. For the reaction to take place, the bonds holding the reactants must be broken so that new bonds of the products are formed, i.e.,
A-B + C-D → A-C + B-D
Old bonds broken: A-B and C-D in reactants
New bonds formed: A-C and B-D in products
The energy required to break one mole of a (covalent) bond is called bond dissociation energy. The SI unit of bond dissociation energy is kJ mole-1.
The higher the bond dissociation energy, the stronger the (covalent) bond.
Bond Dissociation Energies of Some (Covalent) Bonds
Bond Bond Dissociation Energy (kJ mole-1) Bond Bond Dissociation Energy (kJ mole-1) H-H 431 I-I 151 C-C 436 C-H 413 C=C 612 O-H 463 C≡C 836 C-O 358 N≡N 945 H-Cl 428 N-H 391 H-Br 366 F-F 158 C-Cl 346 Cl-Cl 239 C-Br 276 Br-Br 193 C-I 338 H-I 299 O=O 497 Si-Si 226 C-F 494 The molar enthalpy of reaction can be calculated from the bond dissociation energies by:
- Adding the total bond dissociation energy of the reactants (endothermic process / +∆H) and total bond dissociation energy of the products (exothermic process / -∆H).
- Subtracting total bond dissociation energy of the reactants from that of the products (exothermic process / -∆H minus endothermic process / +∆H).
Practice Examples: Calculating ∆Hr
H2(g) + Cl2(g) → 2HCl(g)
Working:
Old bonds broken (endothermic process / +∆H):
= (H-H + Cl-Cl) = (+431 + 239) = +670 kJ
New bonds formed (exothermic process / -∆H):
= 2 × (H-Cl) = 2 × (-428) = -856 kJ
∆Hr = (+670 kJ + -856 kJ) = -186 kJ = -186 kJ mole-1
The above reaction has negative ∆H and is therefore exothermic.
The thermochemical reaction is thus:
½ H2(g) + ½ Cl2(g) → HCl(g) ∆Hr = -93 kJ
CH4(g) + Cl2(g) → CH3Cl + HCl(g)
Working:
Old bonds broken (endothermic process / +∆H):
= 4 × (C-H) + Cl-Cl = (4 × 413) + 239 = 1891 kJ
New bonds formed (exothermic process / -∆H):
= 3 × (C-H) + H-Cl + C-Cl = (3 × -413) + (-428) + (-346) = -2013 kJ
∆Hr = (+1891 kJ + -2013 kJ) = -122 kJ mole-1
The above reaction has negative ∆H and is therefore exothermic.
The thermochemical reaction is thus:
CH4(g) + Cl2(g) → CH3Cl(g) + HCl(g) ∆H = -122 kJ
CH2CH2(g) + Cl2(g) → CH3Cl CH3Cl (g)
Working:
Old bonds broken (endothermic process / +∆H):
= 4 × (C-H) + Cl-Cl + C=C = (4 × 413) + 239 + 612 = 2503 kJ
New bonds formed (exothermic process / -∆H):
= 3 × (C-H) + C-C + 2 × (C-Cl) = (3 × -413) + (-436) + 2 × (-346) = -2367 kJ
∆Hr = (+2503 kJ + -2367 kJ) = +136 kJ mole-1
The above reaction has positive ∆H and is therefore endothermic.
The thermochemical reaction is thus:
CH2CH2(g) + Cl2(g) → CH3Cl CH3Cl (g) ∆H = +136 kJ
Note:
- A reaction is exothermic if the bond dissociation energy of the reactants is greater than that of the products.
- A reaction is endothermic if the bond dissociation energy of the reactants is less than that of the products.


(b) Standard Enthalpy/Heat of Combustion ∆H°c
The molar standard enthalpy or heat of combustion (∆H°c) is defined as the energy or heat change when one mole of a substance is burnt in oxygen or excess air at standard conditions.
Burning is the reaction of a substance with oxygen or air. It is an exothermic process producing a large amount of energy in the form of heat.
A substance that undergoes burning is called a fuel. A fuel is defined as a combustible substance which burns in air to give heat energy for domestic or industrial use. Fuels may be solid (e.g., coal, wood, charcoal), liquid (e.g., petrol, paraffin, ethanol, kerosene), or gas (e.g., liquefied petroleum gas (LPG), water gas (CO2/H2), biogas (methane), natural gas (mixture of hydrocarbons)).
To Determine the Molar Standard Enthalpy/Heat of Combustion (∆H°c) of Ethanol
Procedure:
Put 20 cm3 of distilled water into a 50 cm3 beaker. Clamp the beaker. Determine the temperature of the water T1. Weigh an empty burner (empty tin with wick) and record its mass M1. Put some ethanol into the burner. Weigh again the burner with the ethanol and record its mass M2. Ignite the burner and place it below the clamped 50 cm3 beaker. Heat the water in the beaker for about one minute. Put off the burner. Record the highest temperature rise of the water, T2. Weigh the burner again and record its mass M3.
Sample Results:
Volume of water used 20 cm3 Temperature of the water before heating T1 25.0°C Temperature of the water after heating T2 35.0°C Mass of empty burner M1 28.3 g Mass of burner + ethanol before igniting M2 29.1 g Mass of burner + ethanol after igniting M3 28.7 g Sample Calculations:
(a) Calculate ∆T, the change in temperature:
∆T = T2 – T1 = 35.0°C – 25.0°C = 10.0°C
(b) Calculate the mass of ethanol used in burning:
Mass of ethanol used = M2 – M3 = 29.1 g – 28.7 g = 0.4 g
(c) Calculate the number of moles of ethanol used in burning:
Moles of ethanol = mass used / molar mass = 0.4 / 46 = 0.0087 moles
2. Given that the specific heat capacity of water is 4.2 kJ kg-1 K-1, determine the heat produced during the burning.
Heat produced ∆H = mass of water (m) × specific heat capacity (c) × ∆T
= 0.02 kg × 4200 J/kg·K × 10 = 840 J = 0.84 kJ
3. Calculate the molar heat of combustion of ethanol:
Molar heat of combustion ∆Hc = Heat produced / Number of moles of fuel
= 0.84 kJ / 0.0087 moles = 96.55 kJ mole-1
4. List two sources of error in the above experiment:
- Heat loss to the surroundings lowers the practical value of the molar heat of combustion of ethanol. A draught shield tries to minimize this loss by protecting the flame from wind.
- Heat gain by the reaction vessel or beaker lowers ∆T and hence ∆Hc.
5. Calculate the heating value of the fuel:
Heating value = molar heat of combustion / molar mass of fuel = 96.55 kJ mole-1 / 46 g = 2.10 kJ g-1
Heating value is the energy produced when a unit mass or gram of a fuel is completely burnt.
6. Explain other factors used to determine the choice of fuel for domestic and industrial use:
- Availability and affordability: Some fuels are more readily available and cheaper in rural areas than in urban areas.
- Cost of storage and transmission: A fuel should be easy to transport and store safely. For example, LPG is very convenient to store and use, while charcoal and wood are bulky.
- Environmental effects: Most fuels produce carbon(IV) oxide gas as a byproduct after burning. Carbon(IV) oxide is a greenhouse gas that causes global warming. Some fuels produce acidic gases like sulphur(IV) oxide and nitrogen(IV) oxide, which cause acid rain. Internal combustion engine exhausts produce lead vapour from leaded petrol and diesel, which is carcinogenic.
- Ignition point: The temperature at which a fuel must be heated before it burns in air is the ignition point. Fuels like petrol have very low ignition points, making them highly flammable. Charcoal and wood have very high ignition points.
7. Explain the methods used to reduce pollution from common fuels:
- Planting trees: Plants absorb excess carbon(IV) oxide for photosynthesis and release oxygen gas to the atmosphere.
- Using catalytic converters: In internal combustion engines, catalytic converters convert harmful gases like carbon(II) oxide and nitrogen(IV) oxide to harmless carbon(IV) oxide, water, and nitrogen gas by using platinum-rhodium catalysts along the engine exhaust pipes.
Further Practice Calculations
Calculate the heating value of methanol CH3OH given that 0.87 g of the fuel burns in air to raise the temperature of 500 g of water from 20°C to 27°C (C=12.0, H=1.0, O=16.0).
Moles of methanol used = Mass of methanol used / Molar mass = 0.87 / 32 = 0.02718 moles
Heat produced ∆H = mass of water × specific heat capacity × ∆T = 500 × 4.2 × 7 = 14700 J = 14.7 kJ
Molar heat of combustion ∆Hc = Heat produced / Number of moles of fuel = 14.7 kJ / 0.02718 moles = 540.84 kJ mole-1
Heating value = molar heat of combustion / molar mass of fuel = 540.84 / 32 = 16.90 kJ g-1
1.0 g of carbon burns in excess air to raise the temperature of 400 g of water by 18°C. Determine the molar heat of combustion and hence the heating value of carbon (C=12.0).
Moles of carbon used = Mass of carbon used / Molar mass = 1.0 / 12 = 0.0833 moles
Heat produced ∆H = mass of water × specific heat capacity × ∆T = 400 × 4.2 × 18 = 30240 J = 30.24 kJ
Molar heat of combustion ∆Hc = Heat produced / Number of moles of fuel = 30.24 kJ / 0.0833 moles = 363.03 kJ mole-1
Heating value = molar heat of combustion / molar mass of fuel = 363.03 / 12 = 30.25 kJ g-1