FIRST TERM E-LEARNING NOTE
SUBJECT: CHEMISTRY CLASS: SSS2
SCHEME OF WORK
WEEK TOPIC
- The Periodic Table
- Historical Development of the Periodic Table/Periodic Law
- Features of the Periodic Table.
- Periodic Classification into Blocks and Families.
- Families of Elements.
- The Periodic Trend
- The Periodic Properties of Elements: Atomic size, Ionic size,Ionization Energy, Electron Affinity, Electronegativity down the group and across the period.
- Diagonal Relationship in the Properties of Elements in the Periodic Table.
- Types of Reaction/Redox Reactions
- Definition of Oxidation and Reduction.
- Relationship between Oxidation Number and IUPAC Naming.
- Oxidizing and Reducing Agents
- Identification of Oxidizing and Reducing Agents
- Balancing of Redox Equations in Acidic and Alkaline Medium.
- Ionic Theory: Distinguish between:
- Electrovalent and Covalent Compounds.
- Electrolytes and Non-electrolytes.
- Weak and Strong Electrolytes.
- Conductors and Non –conductors.
- Electrolysis
- Meaning of Electrolysis
- Definition of Terms
- Preferential Discharge of Ions during Electrolysis.
- Electrolysis of Specified Electrolytes
- Acidified water
- Brine solution
- Copper (II) tetraoxosulphate (VI) solution.
- Uses of Electrolysis
- Faraday’s Laws of Electrolysis
- Faraday First and Second laws of electrolysis.
- Verification of Faraday’s Laws of Electrolysis.
- Calculations based on Faraday’s Laws.
- Electrochemical Cells
- Electrolytic and Electrochemical Cells with their differences.
- Standard Electrode Potential.
- Drawing and Writing of Cell Diagrams.
- Calculation of e.m.f of a Cell.
- Application of Electrochemical Cells
- Distinction between Primary and Secondary Cells
- Types of Electrochemical Cells.
REFERENCE MATERIALS
- New School Chemistry for Senior Secondary Schools by O.Y Ababio
- New System Chemistry for Senior Secondary Schools by T. Y. Toon et al
- S.S.C.E Past Questions and Answers on Chemistry
- U.T.M.E Past Questions and Answers on Chemistry
WEEK ONE
TOPIC: THE PERIODIC TABLE
CONTENT
- Historical Development of the Periodic Table/Periodic Law.
- Features of the Periodic Table.
- Periodic Classification into Blocks and Families.
- Families of Elements.
THE PERIODIC TABLE
The periodic table is the table which shows the arrangement of elements in the order of increasing atomic number.
HISTORICAL DEVELOPMENT OF THE PERIODIC TABLE
In Chemistry, classification of elements began with Lavoiser in 1787, whose classification was essentially into metals and non-metals. Other scientists like Doberainer, Newland and Lother Meyer also attempted to classify elements based on their properties. In 1869, a Russian scientist, Dmitri Mendeleev prepared the first periodic table where he arranged elements based on the atomic masses of the elements.
Mendeleev development of the periodic table was based on the periodic law. His periodic law stated that the properties of the element varied periodically with their relative atomic masses. He arranged the elements in increasing order of relative atomic mass. With this, elements having similar properties kept recurring at regular intervals or periods forming families of related elements.
With the discovery of the electronic structure of the atoms, it became clear that elements do vary regularly not with their relative atomic mass, but with their atomic number. Hence, in the modern periodic table, classification is based on atomic number, rather than relative atomic mass. Therefore, the modern periodic law states that the properties of elements are periodic function of their atomic number.
FEATURES OF THE PERIODIC TABLE
The modern form of the periodic table is divided into eight vertical columns known as GROUPS and seven horizontal rows known as PERIODS.
GROUPS:
The vertical columns of elements or groups are numbered from I to VIII (or 0). Elements in the same group have the same number of electrons in the valence shell. Hydrogen can be placed in group I or VII because it can donate its one electron like group I elements or accepts electron like group VII elements. But for convenience and because of its simple valence electron, it is placed in group I. In group VIII, which is also group 0, Helium has two electrons while the other elements have eight valence electrons. Besides the eight groups, there are also, the transition groups of elements. These lay between group II and III in the periodic table.
PERIODS:
The horizontal rows of elements or periods are numbered from 1 to 7. Elements in the same period have the same number of electron shells. Among the elements in the period six and seven are the elements of Lanthanides and Actinides series knows as inner transitions metals.
PERIODIC CLASSIFICATION INTO BLOCKS AND FAMILIES
The elements in the periodic table may be divided into blocks according to the orbital their valence electrons are found which is responsible for the positions of the elements. The s-block elements have s-electrons in the outermost energy level, while the p-block has both s and p-electrons. The transition elements contain d-electrons in addition to its s and p-electrons, while the lanthanides and actinides contain f-electrons in addition to the s, p and d electrons.
Element Atomic Number Electronic configuration.
H 1 1s1
He 2 1s2
Li 3 1s2 2s1
Be 4 1s2 2s2
B 5 1s2 2s2 2p1
C 6 1s2 2s2 2p2
N 7 1s2 2s2 2p3
O 8 1s2 2s2 2p4
F 9 1s2 2s2 2p5
Ne 10 1s2 2s2 2p6
Na 11 1s2 2s2 2p6 3s1
Mg 12 1s2 2s2 2p6 3s2
Al 13 1s2 2s2 2p6 3s2 3p1
Si 14 1s2 2s2 2p6 3s2 3p2
P 15 1s2 2s2 2p63s23p3
S 16 1s2 2s2 2p63s2 3p4
Cl17 1s2 2s2 2p6 3s2 3p5
Ar18 1s2 2s2 2p6 3s23p6
K 19 1s2 2s2 2p6 3s23p6 4s1
Ca20 1s2 2s2 2p6 3s23p6 4s2
EVALUATION
1. State the periodic law.
2. Explain the basis on which elements are arranged in the periodic table.
FAMILIES OF ELEMENTS
Elements in the same group may be said to belong to a family since they show similar properties because their atoms have the same number of valence electrons. At the same time, certain properties of the element in the same group show a gradual change with increase in atomic number. Such gradual change of property within a group is known as GROUP TREND.
GROUP I
The group I elements include: Lithium (Li), Sodium(Na), Potassium(K), Rubidium(Rb), Caesium (Cs), and Francium (Fr). They are univalent elements. They properties are as follows:
- They are good reducing agent since they can readily donate one electron to form cation.
- They are metals, thus they are good conductors of electricity and heat.
- They react vigorously with cold water to liberate hydrogen gas and form alkali, hence, they are known as ALKALI METALS. Example2Na(s) + 2H2O(l)→ 2NaOH(aq) + H2(g)
- The oxides of group I elements dissolve in water to give a very strong alkalis. Example
K2O(s) + H2O(l)→ 2KOH(aq)
GROUP II
Beryllium (Be), Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Ba), and Radium (Ra) belong to group II. They are divalent elements and are also known as alkaline earth metals. Their properties include:
- They ionize by donating their two valence electrons; hence they are good reducing agent.
- They are hard metals, ductile malleable and can conduct both electricity and heat.
Beryllium does not react with cold water or steam, magnesium reacts with steam only while calcium reacts slowly with cold water to liberate hydrogen gas.
- Their oxides are insoluble in water except fort calcium oxide which dissolves in water to form an alkali.
CaO(s) + 2H2O(l)→Ca(OH)2(aq)
GROUP III
The group III elements are: Boron (B), Aluminum (Al), Gallium (Ga), Indium (In) and Thallium (Tl). They are trivalent elements. Their properties are:
- They are reducing in nature since they can donate their three electrons to form electrovalent compounds.
- Only aluminum can react with steam at about750oC to liberate hydrogen gas.
- Oxide and hydroxide of aluminum is amphoteric in nature, i.e, they have both acidic and basic properties. Example
Al2O3(s) + 3H2SO4(aq)→ Al2(SO4)2(aq) + 3H2O(l)
2Al(OH)3(s) + NaOH(aq)→NaAl(OH)4(aq)
GROUP IV
Group IV elements include: Carbon (C), Silicon (Si), Germanium (Ge), tin (Sn) and lead (Pb). They form covalent compounds.
- They exhibit two oxidation states: +2 and +4. Due to inert pair effect of electrons in the s-orbital of the valence shell, the +2 oxidation state becomes more prominent down the group.
- Electropositivity increases down the group. Carbon is a non-metal; silicon and germanium are metalloids while tin and lead are metals.
- Carbon does not react with water in any form; but silicon and tin react with steam at red heat to form +4 state oxides and hydrogen.
Si(s) + 2H2O(l)→ SiO2(s) + 2H2(g)
GROUP V
Nitrogen (N), Phosphorus (P), Arsenic (As), Antimony (Sb) and Bismuth (Bi) belong to group V. They have the following properties
- They exhibit oxidation states of -3 and -5.
- They also show group trend. Nitrogen and phosphorus are non-metals; arsenic and antimony are metalloids while bismuth is a metal.
- They electron acceptors, hence they are oxidizing in nature.
- They form oxides that dissolve in water to form acids except nitrogen (I) oxide.
GROUP VI
Elements in group VI include: Oxygen (O), Sulphur (S), Selenium (Se), Tellurium (Te), and Polonium (Po).their properties are as follows:
- They are non-metals and exist as solid at room temperature except for oxygen
- They are electron acceptors and oxidizing in nature.
- They do not react with water in any form. But oxygen and sulphur combine directly with hydrogen to yield water and hydrogen sulphide respectively.
GROUP VII
Elements in this group include: Fluorine (F), Chlorine (Cl), Bromine (Br), Iodine (I) and Astatine (At). They are known as halogens (salt-makers). Their properties include:
- They ionize to form univalent anions.
- They exist as diatomic molecules.
- As electron acceptor, all halogens are good oxidizing agent.
- They exhibit group trend. Fluorine and chlorine are gases, bromine is a liquid and iodine and astatine are solids at room temperature.
GROUP VIII (0)
The elements in group 0 are known as rare or noble gases because they are non-reactive and exist freely as monoatomic molecules in the atmosphere. The elements that belong to this group are: Helium (He), Neon (Ne), Argon (Ar), Krypton (Kr), Xenon (Xe) and Radon (Rn).
TRANSITION ELEMENTS
These are elements found in-between group II and III of the periodic table. The first transition series consists of elements: Scandium (Sc), Titanium (Ti), Vanadium (V), Chromium (Cr), Manganese (Mn), Iron (Fe), Cobalt (Co), Nickel (Ni), Copper (Cu) and Zinc (Zn). Transition elements have the following properties:
1. High tensile strength
2. High melting and boiling points
3. Variable oxidation states
4. Formation of colored ions
5. Formation of complex ions
6. Paramagnetic in mixture
7. Catalytic ability
LANTHANIDES (RARE EARTH ELEMENTS): These are found in period six. This series begins with Lanthanum (La), and ends with Lutetium (Lu). They are altogether 15 and resemble one another greatly.
ACTINIDES AND THE ARTIFICIAL ELEMENTS: The actinides are similar to the Lanthanides. They are found in the seventh period, which starts with Actinium (Ac) and ends with Lawrencium, (Lr). The famous Uranium is in this group.
The elements with atomic number from 93 to 103 are known as the artificial elements. This is because they do not occur naturally but were formed during nuclear reactions.
GENERAL EVALUATION/REVISION
- State the division of modern periodic table
- Hydrogen can be placed in group I or VII. Explain
- Write the electronic configuration of the following element and state the group and period to which they belong: 6C, 11Na, 14S, 18Ar
- State the number of unpaired electron in each of the following atom/ion: 12Mg2+, 16S2-17Cl
- Name ten laboratory apparatuses and state their uses
READING ASSIGNMENT: New School Chemistry for Senior Secondary School by O.Y.Ababio, pages 141-143, 150-154
WEEKEND ASSIGNMENT
SECTION A: Write the correct option ONLY
- The vertical columns in the periodic table are referred to as A. periods B. groups C. rows D. cells.
- The arrangement of elements in the periodic table is based on their
A. atomic number B. molecular mass C. Avogadro’s number D. Dalton’s theory.
- One of the following elements in NOT a halogen A. chlorine B. iodine C. astatine
D. antimony
- Physical and chemical properties of elements are periodic function of atomic number is a state of A. gas law B. periodicity C. periodic law D. regular variation.
- The lightest element in the periodic table is A. Helium B. Lithium C. Hydrogen
D. Argon.
SECTION B
- The electronic configuration of five elements represented by the letters P, Q, R, S and T are indicated below:
P- 1s2 2s2 2p2
Q- 1s2 2s2 2p4
R – 1s2 2s2 2p6
S – 1s2 2s2 2p6 3s2
T – 1s2 2s2 2p6 3s2 3p5
Without identifying the elements, state which of them
- belongs to group VI in the periodic table
- readily ionize by gaining electron
- contains two unpaired electrons in the ground state
- does not participate readily in chemical reactions
- is an s-block element
- Copy and complete the table below
Particle | Number of protons | Number of electrons | Number of neutrons |
1H1 | 1 | 1 | |
27Al133+ | 14 | ||
16O82- | 8 |
WEEK TWO
TOPIC: THE PERIODIC TRENDS
CONTENT
- The Periodic Properties of Elements: Atomic size, Ionic size, Ionization Energy, Electron Affinity, Electronegativity down the group and across the period.
- Diagonal Relationship in the Properties of Elements in the Periodic Table.
PERIODICITY
Periodicity with respect to the periodic table is defined as the variation in the properties of elements in a regular pattern both down the groups and across the periods.
REASONS FOR PERIODIC VARIATION IN PROPERTIES
Two opposing factors are responsible for the variation in properties of elements. These are
1. Effect of increasing positive nuclear charge.
2. Screening effect of inner shell electrons.
EFFECT OF NUCLEAR CHARGE AND INNER SHELL ELECTRONS
1. Nuclear charge exerts force of attraction on the electrons towards the nucleus. This force of attraction increases as a result of increasing nuclear charge across periods.
2. Inner shell electrons screen the nuclear charge from exerting its force of attraction outwards to outer electrons. Screening effect is constant across each period but increase down the groups.
The overall effects of these are:
- Effect of increasing nuclear charge overpowers the screening effect of
inner shell electrons across a period.
b. Screening effect of inner shell electrons overpowers the effect of increasing nuclear charge down the groups.
These effects are the reasons for periodic variations in properties of elements.
ATOMIC PHYSICAL PROPERTIES OF ELEMENTS THAT SHOW PERIODIC VARIATION
- ATOMIC RADIUS: Atomic radius is one half the distances between two covalently bonded atoms. For electrovalent compounds, ionic radius is a measure of the distance between the centre of the ion and the centre of its nearest neighbour of opposite charge.
- IONIZATION ENERGY: Ionization energy is the energy required to remove one mole of electrons from one mole of gaseous atom to produce one mole of gaseous ions.
- ELECTRONEGATIVITY: Electronegativity is the power of an atom of an element to attract electrons to itto become negatively charged.
- ELECTROPOSITIVITY: Electropositivity is the power of the atom of an element to lose electron and become positively charged.
- ELECTRON AFFINITY: Electron affinity is the energy change which accompanies the addition of one mole of electrons to one mole of gaseous atom of an element to form negatively charge ions.
EVALUATION
- State five atomic physical properties of elements.
- Define two of the stated atomic physical properties
TRENDS IN ATOMIC PROPERTIES
1. Atomic radius decreases across the periods from left to right while it increases down the groupsfrom top to bottom.
2. Ionization energy increases across the periods left to right while it decreases down the groups from top to the bottom.
3. Electronegativity increases from left to right across each period while it decreases down the groups from top to bottom.
4. Electropositivity decreases from left to right across the period while it increases down the groups from top to bottom.
5. Electron affinity decreases from left across each period while it increasesdown the groups.
6.Ionic radius decreases for metal across the periods and increase for non – metals.
Ionic radius generally increases down the groups and decreases across the periods.
NOTE
Cations formed by electron loss are smaller in size than the neutral atom from which they are formed and anions formed by electron gain are larger in size than the neutral atoms that form them. Also, the larger the charge on a cation, the smaller the ionic radius and the larger the charge on the anion, the larger the ionic radius.

EVALUATION
State the trends of the following across the periods and down the groups
1. Ionization energy
2. Atomic radius.
VARIATION IN OTHER PHYSICAL PROPERTIES
1. MELTING POINT AND BOILING POINT:
The melting point is the temperature at which a solid begins to melt at a particular pressure. The boiling point is the temperature at which a liquid begins to boil at a particular pressure.
The melting and boiling points of elements increase from groups I to III across each period with group IV elements having the highest value and decrease from group V to 0. The melting and boiling point decrease down the groups from group I to IV and increase down the groups from group V to 0. To account for this trend, in the properties across period, there is an increase in metallic bond from group I to III and a lot of energy is needed to break this bond. The presence of giant covalent lattice in group IV elements results in the high melting and boiling points. From group V to 0, there is decrease in the weak intermolecular force of attraction among the molecules of the atoms of elements. Thus, there is a decrease in melting and boiling point.
2. ELECTRICAL AND THERMAL CONDUCTIVITY: These properties of elements decrease across the period and increase down the group. Thus, metals are good electrical and thermal conductors while non- metals are poor electrical and thermal conductors.
EVALUATION
1. Explain variation of ionic radius in the periodic table.
2. Explain the variation of melting and boiling points in the periodic table.
VARIATION IN CHEMICAL PROPERTIES
The chemical properties of elements also exhibit periodic variation. This is also shown in the properties of the compounds of the elements.
GROUPS: Elements in the same group have similar chemical properties because they have the same number of valence electrons. The four groups of elements, which show great similarity in their chemical properties, are
- Group 1 element or the Alkali metals.
- Group II elements or the Alkaline earth metal
- Group VII elements or the Halogens
- Group 0 elements or the noble or rear gases
PERIOD:Elements in the same period do not exhibit similar chemical properties. Chemical properties change across the periods.
The following chemical properties shall be considered
1. Chemical reactivity
2. Compound thermal stability
3. Ease of formation of ions
4. Metallic properties and Non – metallic properties
1. Chemical reactivity decrease from group I – III for metals and increase from group IV to VII for non-metals across the periods while it increase down groups for metal and decreases down groups for non-metals.
2. Compound thermal stability decrease from Group I to III for metals and increases from IV to VII for non- metals across the period while it increases down each group for metals and decreases for non-metal respectively.
3. Ease of formation of ions decreases from Group I – III across the periods for metals and increases from IV to VII for non-metals while it increases down the group for metals and decreases down the groups for non-metals
4. Metallic property decreases across the periods and increases down groups while Non metallic property increases across periods and decreases down groups.
EVALUATION
1. State the valence electron of each of the group of elements:
- Alkali metals (b) Halogens (c) Alkaline earth metals
2. Briefly explain the variation in the following chemical properties:
(a) Chemical reactivity (b) Ease of formation of ions (c) Metallic property
DIAGONAL RELATIONSHIP IN THE PROPERTIES OF ELEMENTS IN THE PERIODIC TABLE
Diagonal relationship is said to exist between certain pairs of diagonally adjacent elements in the second and third periods of the periodic table example Lithium and Magnesium, Beryllium and Aluminum, Boron and Silicon.
It is found that the chemistry of a first-row (second period) element often has similarities to the chemistry of the second-row (third period) element being one column to the right of it in the periodic table. Thus, the chemistry of Lithium has similarities to that of Magnesium, the chemistry of Beryllium has similarities to that of Aluminium, and the chemistry of Boron has similarities to that of Silicon. These are called diagonal relationships. (It is not as noticeable after Boron and Silicon.) The reasons for the existence of diagonal relationships are not fully understood, but charge density is a factor. For example, Li+is a small cation with a +1 charge and Mg2+is somewhat larger with a +2 charge, so the charge density on each ion is roughly the same. Let’s consider the Li–Mg pair:(under room temperature and pressure)
- Lithium and Magnesium form only normal oxides whereas sodium forms peroxide and metals below sodium, in addition, form superoxides.
- Lithium is the only Group I element which forms a stable nitride, Li3N.Magnesium, as well as other Group II elements, also forms nitrides.
- Lithium trioxocarbonate (IV) and Lithium fluoride are sparingly soluble in water. The corresponding Group II salts are insoluble.
- Chlorides of both Li and Mg are deliquescent (absorb moisture from surroundings) and are soluble in ethanol. Also, Lithium chloride, like magnesium chloride (MgCl2.6H2O) separates out from solutions as hydrated crystal LiCl.2H2O.
GENERAL EVALUATION/REVISION
- Define the following terms: Ionization energy, Electronegativity, Electron affinity
- Describe the variation in each of the terms defined in (1) above across a period and down a group
- Arrange the following ions in order of increasing size: Na+, Ca2+ and Al3+. Give reason for your answer.
- Define physical and chemical change giving two examples each.
READING ASSIGNMENT
New School Chemistry for Senior Secondary School by O. Y. Ababio, pages 144-150
WEEKEND ASSIGNMENT
SECTION A: Write the correct option ONLY
- The complete modern periodic table consists of A. 7 Groups and 8Periods
B. 8 Groups and 4Periods C. 8Groups and 8 Periods D. 8 Groups and 7 Periods
2. Which of the following is correct? A. Nuclear charge increases both across the periods and down the groups. B. Number of electron shells is constant across each group C. Force of attraction of the nuclear charge on the valence electrons decreases across theperiods and increases down the groups. D. Number of inner shell electrons is constant across each group.
3. All of the following are atomic properties except A. Atomic radius B. Ionization energy. C. Melting point D. Electron Affinity
4. The atom of an element is represented by 3115 X, which of the following is not correct about element X? ItA. belongs to group V and period 3 B. the properties of X are functions of the number 15 C. is a non metalD. the properties of X are function of number 31
5. Ionization energy increases from left to right across each period. Which of the following cannot be a reason for this variation? A. Effect of increasing nuclear charge overpowers the screening effect of inner shell electrons.
B. There is gradual increase in amount of energy required to remove a valence electron from the atoms of the elements.
C. Nuclear force of alteration on valence electrons increases as the nuclear charge increasesD. none of the above
SECTION B
1a. Give the reason why atomic radius increases down a group in the periodic table but decreases from left to right in a period.
- Arrange the following in order of increasing ionic size: P3-, Cl– and S2-. Give reason for your answer
2a.State the variation in the following atomic properties across a period and down a group: (i) Ionization energy (ii) Electronegativity
b. What is diagonal relationship?
WEEK THREE
TOPIC: TYPES OF REACTION
CONTENT
TYPES OF REACTIONS
Chemical reactions are changes in which some new chemical substances are formed. Chemical reactions can be grouped into:
- Combination reaction: This involves the combination of two or more substances to form a single compound. Example
PbO2(s) + SO2(g)→ PbSO4(s)
- Displacement reaction: In displacement reaction, one element or radical replaces another element or radical in a compound. Example
Zn(s) + CuSO4(aq)→ ZnSO4(aq) + Cu(s)
- Double decomposition: This reaction involves the reactants decomposing to form new substances by an exchange of radicals. Example
AgNO3(aq) + NaCl(aq) →AgCl(s) + NaNO3(aq)
- Reversible reaction: A reversible reaction is a reaction which the products can react to give the reactants. Example
CaCO3(s) + H2O(l) + CO2(g) Ca(HCO3)2(aq)
- Thermal dissociation: In thermal dissociation, each molecule of a substance dissociates into two simpler molecules or atoms on the application of heat. Unlike thermal decomposition, thermal dissociation is reversible. Example
NH4Cl(s) NH3(g) + HCl(g)
- Catalytic reaction: Any chemical reaction which uses a catalyst is a catalytic reaction.E.g
H2(g) + I2(g) → 2HI(g)
A catalyst is a substance which alters the rate of a chemical reaction, but itself remains chemically and quantitatively unchanged at the end of the reaction.
CHARACTERISTICS OF A CATALYST
- It alters the rate of a chemical reaction.
- It remains unchanged in chemical nature and mass at the end of a reaction.
- It is specific in action.
- It cannot start a reaction.
- It does not affect the types of products formed in a reaction.
- It has no effect on the equilibrium of a reversible reaction.
- The effect of a solid catalyst is improved by increasing its surface area.
TYPES OF CATALYSTS
Catalysts may be inorganic or organic substances. Inorganic catalysts can be grouped into positive and negative catalysts. Organic catalysts are known as enzymes and are produced by living cells.
A catalyst which increases the rate of a reaction is a positive catalyst. An example is manganese (IV) oxide which speeds up the decomposition of potassium trioxochlorate (V).
2KClO3(s) →2KCl(s) + 3O2(g)
A catalyst which decreases the rate of a reaction is a negative catalyst. Example, tetraoxophosphate (V) acid decreases the rate of decomposition of hydrogen peroxide.
2H2O2(l) → 2H2O(l) + O2(g)
Promoters and Inhibitors: A promoter is a substance that influences the rate of a reaction by affecting the efficiency of the catalyst. Example, aluminum oxide is added to iron catalyst used in the Haber process for the production of ammonia.
An inhibitor is a substance that decreases the efficiency of a catalyst. Example, hydrogen cyanide and mercury salts inhibit important enzymes in our bodies.
TYPES OF CATALYSIS
Homogeneous catalysis: In this reaction, the catalyst, reactants and products are all in the same phase. Example
2SO2(g) + O2(g) → 2SO3(g)
Heterogeneous catalysis: For heterogeneous catalysis, the reactants, products and catalysts are in different phases. Example
H2(g) + I2(g) → 2HI(g)
EVALUATION
- State what type of reaction is represented by each of the following equations:
- 2KBr(aq) + Cl2(g)→ 2KCl(aq) + Br2(l)
- 2Pb(NO3)2(s)→ 2PbO(s) + 4NO2(g) + O2(g)
- KNO3(aq) + H2SO4(aq)→ KHSO4(aq)
- Define the term Catalyst and give five characteristics of a catalyst.
OXIDATION AND REDUCTION
DEFINITION OF OXIDATION AND REDUCTION
The following definitions, have been used for the oxidation and reduction processes
- In terms of Oxygen: Oxidation is defined as the addition of oxygen to a substance while reduction is defined as the removal of oxygen from a substance. Examples


 Reduction
Reduction 2Mg(s) + O2(g) 2MgO(s)
2Mg(s) + O2(g) 2MgO(s) 
 R.A O.A
R.A O.A Oxidation
Oxidation

 R.AReduction
R.AReduction
 C(s) + 2ZnO(s) CO2(s) + 2Zn(s)
C(s) + 2ZnO(s) CO2(s) + 2Zn(s) 
 O.A
O.AOxidation
- In terms of Hydrogen: Oxidation is defined as the removal of hydrogen from a substance while reduction is the addition of hydrogen to a substance. Example
Reduction


 R.A
R.A H2S(g) + Cl2(g) 2HCl(g) + S(s)
H2S(g) + Cl2(g) 2HCl(g) + S(s) 

 O.A
O.AOxidation
- In terms of electronegative elements:Oxidation is defined as the addition of electronegative element to a substance while reduction is the removal of electronegative element from a substance. Example
Reduction


 R.A
R.A
 Na (s) + Cl2(g) NaCl(s)
Na (s) + Cl2(g) NaCl(s)

 O.A
O.A
Oxidation

 R.A Reduction
R.A Reduction
 C(s) + 2ZnO(s) CO2(g) + 2Zn(s)
C(s) + 2ZnO(s) CO2(g) + 2Zn(s)

 O.A
O.A
Oxidation
- In term of electron transfer: Oxidation is defined as loss of electron from substances while reduction is defined as gain of electron by substance. Example
Reduction

 O.A
O.A
 FeCl2 (s) + Cl2 (g) FeCl3 (s)
FeCl2 (s) + Cl2 (g) FeCl3 (s)


 R.A
R.A
Oxidation
EVALUATION
- Define oxidation and reduction in terms of
- Hydrogen (b) Oxygen
- Give the modern definition of Oxidation and Reduction
RELATIONSHIP BETWEEN OXIDATION NUMBER AND IUPAC NAMING
The oxidation number or state of an element in a neutral compound or radical (ion) is defined as the numerical electrical charge it appears to have as determined by a set of arbitrary rules.
RULES OF DETERMINING OXIDATION NUMBERS
1. The oxidation number of all uncombined element is zero e.g Na, Cl
2. The oxidation number of an ion consisting of a single element is the same as the ionic charge on it. E.g. oxidation number of Na+ is +1, Cl– is -1
3. The oxidation number of an ion consisting of more than one element (radical) is the algebraic sum of the oxidation number of all the elements in the ion.
Example for OH–
[Oxidation no. of O] + [oxidation no. of H] = [oxidation no of OH–]
(-2) + (+1) = -1
The oxidation number of NH4+ is +1,
4. The oxidation number of a neutral compound is zero and it is the algebraic sum of the oxidation number of all the elements in the compound.
Example: MgCl2= 0
[Oxidation no. of Mg] + 2[oxidation of no.Cl] = [ oxidation number of MgCl2]
+2 + 2 x (-1) = +2 -2 = 0
EXAMPLE
- Find the oxidation number of the manganese atom in KMnO4 and give the IUPAC name the compound.
Solution:
KMnO4 = 0
(+1) + Mn + 4(-2) =0
+1 +Mn -8=0
Mn – 7 =0
Mn = +7
The oxidation number (state) of manganese in KMnO4 is + 7.
Hence the IUPAC name of the compound is Potassium tetraoxomanganate (VII)
- Find the oxidation number of chlorine in NaClO and give the IUPAC name of the compound.
Solution:
NaClO = 0
(+1) + Cl + (-2) = 0
Cl -2 + 1 = 0
Cl -1 = 0
Cl = +1
The oxidation number of chlorine in NaClO is +1.
Thus, the IUPAC name of the compound is Sodium oxochlorate (I)
IDENTIFYING REDOX REACTIONS USING OXIDATION NUMBER
Redox reactions can be identified with the aid of oxidation number. For a redox reaction, oxidation number of an element on the reactant side is different on the product side. There must be changes in oxidation numbers.
 Example: C + ZnO CO2 + Zn Redox
Example: C + ZnO CO2 + Zn Redox
0 +2 +4 0
 NaOH + HCl NaCl + H2O Not Redox
NaOH + HCl NaCl + H2O Not Redox
(+1)(-2) (+1) (+1)(-1) (+1)(-1) (+1×2) (-2)
GENERAL EVALUATION/REVISION
- Show that the following reactions are redox reaction
- 2H2S(g) + SO2(g)→3S(s) + 2H2O(l)
- 2K(s) + 2H2O(l)→ 2KOH(aq) + H2(g)
- 2FeCl(aq) + Cl2(g)→ 2FeCl3(aq)
2. What is the oxidation number of chromium in K2Cr2O7? Give the IUPAC name of the compound.
3. Define oxidation and reduction in modern terms
4. Define radicals and give TWO examples
5. What is valency? Give the valency of the following elements: sodium, phosphorus, carbon, sulphur, and aluminum.
READING ASSIGNMENT: New School Secondary for Senior Secondary School by O. Y. Ababio, pages 184-192
WEEKEND ASSIGNMENT
SECTION A: Write the correct option ONLY
- Major difference between thermal dissociation and thermal decomposition is that
A. Thermal dissociation is not reversible B. thermal decomposition is reversible C. thermal dissociation is reversible D. thermal decomposition is not reversible.
- Removal of hydrogen from a substance is A. oxidation B. reduction C. oxidizing agent
D. reducing agent
- The oxidation number of uncombined element is A. 2 B. 4 C. –1 D. 0.
-
 H2S + O2 S + H2O
H2S + O2 S + H2O
In the above equation of reaction, the oxidation number of sulphur changes from
- –2 to 0 B. +2 to –1 C. 0 to +2 D. +1 to -2
- What is the oxidation number of chromium in sodium heptaoxodichromate (VI)? A. +3 B. +12 C. +6 D. +2
SECTION B
- Define oxidation and reduction in terms of electron transfer.
- Determine the oxidation number of Chromium in K2Cr2O7.
WEEK FOUR
TOPIC:OXIDIZING AND REDUCING AGENTS
CONTENT
- Identification of Oxidizing and Reducing agents
- Balancing of Redox equation in Acidic and Alkaline medium
OXIDIZING AND REDUCING AGENTS
An oxidizing agent is defined as a substance which loses oxygen or electronegative element to another substance. Or an oxidizing agent is a substance which gains hydrogen from a substance. Or an oxidizing agent is a substance which gains electron from a substance. Consider the reaction below
 C(s) + ZnO(s) CO2(g) + Zn(s)
C(s) + ZnO(s) CO2(g) + Zn(s)
ZnO is the oxidizing agent because it loses oxygen to C.
A reducing agent is defined as substance, which removes and accepts oxygen from other substances. Or a reducing agent is defined as a substance, which removes and accepts electronegative element from another substance. Or a reducing agent is defined as a substance which loses and donates electron to another substance. From the reaction above, C is the reducing agent because it removes and accepts oxygen from ZnO.
In an oxidation and reduction reaction, the oxidizing agent is the reduced species while the reducing agent is the oxidized species.
NOTE: An oxidizing agent accepts electron, is reduced and its oxidation number decreases while a reducing agent donate electron, is oxidized and its oxidation number increases.
IDENTIFICATION OF OXIDIZING AND REDUCING AGENTS
TESTS FOR OXIDIZING AGENTS: The presence of an oxidizing agent can be detected using any of the following reagents.
- Acidified potassium iodide, KI with starch
- Sulphur (IV) oxides, SO2 with acidified Barium trioxonitrate (V) solution
- Iron (II) Chloride solution (FeCl2)
- Hydrogen sulphide gas (H2S)
SUMMARY OF TEST
S/NO | TEST | OBSERVATION | INFERENCE |
1 | O.A + FeCl2(aq) | Green colour of Fe2+ solution turns to reddish- brown of Fe3+ | O.A is present |
2 | O.A + H2S(g) | Formation of yellow deposits of sulphur | O.A is present |
3a. b. | O.A + acidified KI Red- brown solution + starch | Reddish – brown coloration produced. Iodine is liberated. Reddish- brown turns dark blue. The iodine reacts with the starch | O.A is present |
4 | O.A + SO2(g) + dilute HNO3(aq) + Ba(NO3)2(aq) | White precipitate of insoluble BaSO4 is formed | O.A is present |
TEST FOR REDUCING AGENTS: Reducing agent is detected in the laboratory using any of the following reagents.
1. Acidified Potassium tetraoxomanganate (VII)
2. Acidified Potassium heptaoxodichromate (VI)
S/N | TEST | OBSERVATION | INFERENCE |
1 | R.A + acidified KMO4 | Purple solution of KMnO4 turns colorless on addition of R.A | R.A is present |
2 | R.A + acidified K2Cr2O7 | Orange solution of K2Cr2O7 turns green solution addition of R.A | R.A is present |
Common oxidizing agents are: concentrated HNO3, H2SO4, KMnO4, K2Cr2O7, O2, Cl2 etc.
Common reducing agents are: concentrated HCl, pure metals, carbon, H2, SO2, H2S, etc.
EVALUATION
1. Define Oxidizing agent and Reducing agent in terms of electron transfer
2. Describe one test each for identifying an Oxidizing agent and a Reducing agent
BALANCING OF REDOX EQUATIONS
Redox equations are balanced by first considering the two half equations involved in such reaction. Steps involved are
1. Identify the oxidizing and reducing agents and deduce expected products.
2. Write the half equations for oxidation and reduction. Balance the atoms and charges for each equation.
3. Make sure that the electrons loss in the oxidation half equation is balanced by the electrons gain in the reduction half equation.
4. Combine the halves equations to eliminate the electrons and get the overall redox equation.
EXAMPLE 1: Write a balanced ionic equation for the redox reaction between potassium tetraoxomanganate(VII) and Iron (II)tetraoxosulphate(VI)in acidic medium.
SOLUTION:
 O.A MnO4–
O.A MnO4–
 R.A Fe2+
R.A Fe2+
OXIDATION HALF EQUATION
 Fe2+ Fe3+ + e–
Fe2+ Fe3+ + e–
REDUCTION HALF EQUATION
 MnO4– + H+ Mn2+ + H2O
MnO4– + H+ Mn2+ + H2O
BALANCED HALF EQUATIONS
 5Fe2+ Fe3+ + 5e–
5Fe2+ Fe3+ + 5e–
 MnO4– + 8H++ 5e– Mn2+ + 4H2O
MnO4– + 8H++ 5e– Mn2+ + 4H2O
COMBINED EQUATION
 5Fe2+ + MnO4– +8H+ + 5e– Fe3++ 5e– + Mn2+ +4H2O
5Fe2+ + MnO4– +8H+ + 5e– Fe3++ 5e– + Mn2+ +4H2O
The electrons on both sides of the equation cancel out and the overall equation is
 5Fe2+ + MnO4– + 8H+ Fe3+ + Mn2+ + 4H2O
5Fe2+ + MnO4– + 8H+ Fe3+ + Mn2+ + 4H2O
EXAMPLE 2: Write a balanced equation for the following reaction in basic medium
 Cr3+ + BrO– CrO42- + Br–
Cr3+ + BrO– CrO42- + Br–
SOLUTION:
 O.A BrO–
O.A BrO–
 R.A Cr3+
R.A Cr3+
OXIDATION HALF EQUATION
 Cr3+ CrO42-
Cr3+ CrO42-
 Balancing of atoms: Cr3+ + 8OH– CrO42- + 4H2O
Balancing of atoms: Cr3+ + 8OH– CrO42- + 4H2O
 Balancing of charges: Cr3+ + 8OH– CrO42- + 4H2O + 3e–
Balancing of charges: Cr3+ + 8OH– CrO42- + 4H2O + 3e–
REDUCTION HALF EQUATION
 BrO– Br–
BrO– Br–
 Balancing the atoms: BrO– + H2O Br– + 2OH–
Balancing the atoms: BrO– + H2O Br– + 2OH–
 Balancing the charges: BrO– + H2O + 2e– Br– + 2OH–
Balancing the charges: BrO– + H2O + 2e– Br– + 2OH–
But electron lost in the oxidation half must equal electron gained in the reduction half equation.
Multiplying the oxidation half equation by 2 and the reduction half equation by 3 gives
BALANCED HALF EQUATIONS
 2Cr3+ +16OH– 2CrO42- + 8H2O + 6e–
2Cr3+ +16OH– 2CrO42- + 8H2O + 6e–
 3BrO– + 3H2O + 6e– 3Br– + 6OH–
3BrO– + 3H2O + 6e– 3Br– + 6OH–
COMBINED EQUATION
2Cr3+ + 16OH– 
 3BrO– + 3H2O + 6e– 2CrO42- + 8H2O + 6e– 3Br– + 6OH–
3BrO– + 3H2O + 6e– 2CrO42- + 8H2O + 6e– 3Br– + 6OH–
The electrons on both sides of the equation cancel out and the overall equation is
 2Cr3+ + 16OH– 3BrO– + 3H2O 2CrO42- + 8H2O + 3Br– + 6OH–
2Cr3+ + 16OH– 3BrO– + 3H2O 2CrO42- + 8H2O + 3Br– + 6OH–
GENERAL EVALUATION/REVISION
1. Determine the oxidation number of
(a) Fe in Fe2O3 (b) Cu in [Cu(NH3)4]2+
2. Name the following compounds
- H2CO3 (b) KMnO4
3.The compound Na2S is called ————–
4.The IUPAC name of NaHSO4 is————-
 5.Balance the following redox equation: I– + MnO4–IO3– + MnO2in basic medium
5.Balance the following redox equation: I– + MnO4–IO3– + MnO2in basic medium
READING ASSIGNMENT
New School Chemistry for Senior Secondary School by O. Y. Ababio, pages 193-196
WEEKEND ASSIGNMENT
SECTION A: Write the correct option ONLY
1 What is the value of x in the following equation?
 Cr2O72-+ 14H+ + xe- 2Cr3+ + 7H2 O
Cr2O72-+ 14H+ + xe- 2Cr3+ + 7H2 O
A. 1 B. 6 C. 8 D. 12
2 In which of the following is the oxidation number of sulphur equal to -2?
A. S8 B. H2S C. SO2D. SO3.2-
3 Which species undergoes reduction in the reaction represented by the equation below?
 H2S(g) + 2FeCl3(aq) S(s) +2HCl(aq) + 2FeCl2(aq)
H2S(g) + 2FeCl3(aq) S(s) +2HCl(aq) + 2FeCl2(aq)
A. Fe3+ B. H2S C. Cl– D. S
 4 Cr2O72- + 6Fe2++ 14H+ 2Cr 3+ + 6Fe3+ + 7H2O In the equation above, the oxidation number of chromium changes from
4 Cr2O72- + 6Fe2++ 14H+ 2Cr 3+ + 6Fe3+ + 7H2O In the equation above, the oxidation number of chromium changes from
A. +7 to +3 B. +6 to +3 C. -6 to +3 d. -2 to +6
5 When SO2 is passed into a solution of acidified potassium heptaoxodichromate(VI) (K2Cr2O7), the solution turns A. green B. orange C. purple d. yellow
SECTION B
1. Determine the oxidation state of P in each of the following structure
a. POCl3 b. PH3
 2. Balance the following redox equation: Cr2O72- + SO2 Cr3+ + SO42- in acidic medium
2. Balance the following redox equation: Cr2O72- + SO2 Cr3+ + SO42- in acidic medium
WEEK FIVE
TOPIC: IONIC THEORY
CONTENT
Distinguish between:
- Electrovalent and Covalent Compounds.
- Electrolytes and Non-electrolytes.
- Weak and Strong Electrolytes.
- Conductors and Non –conductors.
IONIC THEORY
The ionic theory was first presented by Arrhenius to explain electrolysis. Ionic theory proposed that when an electrolyte is melted or dissolved in water, some if not all, the molecules of the substance dissociate into freely moving charged particles called ions. The process of dissociation into ions is known as ionization.
The metallic ions, ammonium ions, NH4+, and hydrogen ions, H+, are positively charged while the nonmetallic ions and hydroxide ions are negatively charged. When electric current is passed through an electrolyte, the free ions lose their random movement. The positive ions become attracted to the cathode (negative electrode) and are known as cations (i.e cathode ions). The negative ions move towards the anode (positive electrode) and are called anions. (i.e anode ions). Therefore, the current through electrolyte is carried by the movement of ions to the electrodes, and not by flow of electrons in the electrolyte.
Arrhenius version of the ionic theory has been modified and replaced by the modern ionic theory. The modern ionic theory propose that an electrolytes consists of oppositely charged ions even in the solid state and such ions are pulled away from one another either as a result of the heat applied when the solid melts or with the help of the solvent molecules when the solid dissolves.
 ARRHENIUS THEORY: NaCl(s) Na+(aq) + Cl–(aq)
ARRHENIUS THEORY: NaCl(s) Na+(aq) + Cl–(aq)
 MODERN THEORY: Na+Cl–(s) Na+(aq) + Cl–(aq)
MODERN THEORY: Na+Cl–(s) Na+(aq) + Cl–(aq)
ELECTROVALENT AND COVALENT COMPOUND
Electrovalent compounds are formed by complete transfer of an electron(s) from a metal to a non-metal and they are made up of ions while covalent compounds are formed by the sharing of electrons between the two atoms combining together and they are made up of molecules.
CHARACTERISTICS OF ELECTROVALENT COMPOUNDS
- Electrovalent compounds are usually crystalline solids at room temperature.
- Electrovalent compounds are usually soluble in polar solvents like water but insoluble in non-polar solvents like tetrachloromethane.
- Electrovalent compounds generally have high melting and boiling points.
- Electrovalent compounds are good conductors of electricity in the molten state and in aqueous solutions but insulators in the solid state.
CHARACTERISTICS OF COVALENT COMPOUNDS
- Covalent compounds are usually liquids or gases.
- Covalent compounds are soluble in non-polar solvents like benzene or carbon tetrachloride and insoluble in polar solvents like water.
- Covalent compounds generally have low melting and boiling points.
- Covalent compounds are bad conductors of electricity.
EVALUATION
- Explain the ionic theory.
- Distinguish between electrovalent compounds and covalent compounds.
ELECTROLYTES AND NON-ELECTROLYTES
An electrolyte is any salt or molecule that ionizes when dissolved in solution and the solution can conduct electricity while a non-electrolyte does not dissociate in solution and does not conduct electricity. Electrolytes can conduct electricity because when a salt dissolves, its dissociated ions can move freely in solution, allowing the flow of -charges.
Examples of electrolytes are: molten or aqueous sodium chlorine, mineral acids like: tetraoxosulphate (VI) acid, hydrochloric acid, potassium hydroxide etc.
Examples of non-electrolytes are: ethanol, benzene, sugar, urea, etc.
WEAK AND STRONG ELECTROLYTES
Strong electrolyte is one which ionizes completely in solution and conduct electric current readily example: tetraoxosulphate (VI) acid, sodium hydroxide, sodium chloride etc; while weak electrolyte is one which ionizes partially in solution and does not conduct electric current readily example ethanoicacid, aqueous ammonia, water and so on.
The conductivity of strong electrolytes decreases slightly with increasing concentration; however the conductivity of weak electrolytes increases with decreasing concentration.
CONDUCTORS AND NON–CONDUCTORS
Any substance that allows the passage of electricity through it is called a conductor; while any substance that does not allow the passage of electricity is known as non-conductor or insulator.
Conductors are mostly metals. Glass, porcelain, plastic, rubber and diamond areexamples of non–conductivematerials
GENERAL EVALUTION/REVISION
- Distinguish between conductor and non-conductor giving examples.
- Differentiate between strong electrolyte and weak electrolyte giving examples.
- State THREE differences between electrovalent and covalent compounds.
- Write the electronic configuration of the following atom/ion: S2-, Al3+, Fe, Cl and Ar.
- Two isotopes of the element Z with mass numbers 18 and 20 are in the ratio of 1:2. Determine the relative atomic mass of Z.
READING ASSIGNMENT
New School Chemistry for Senior Secondary School (6th edition) by O. Y. Ababio, pages 200-201
WEEKEND ASSIGNMENT
SECTION A: Write the correct option ONLY
- Which of the following liquids is a good conductor of electricity? A. Methylbenzene B. Deionized water C. Mercury D. Sucrose solution
- Potassium chloride cannot conduct an electric current in the solid state because it A. does not contain mobile ions. B. is very soluble in water. C. is an electrovalent compound D. is a neutral salt.
- What happens to the conductivity of a strong electrolyte as its concentration reduces? It A. increases B. decreases C. is unaffected D. reduces to zero
- The current carriers that are responsible for the conductance of electrolytes are A. hydrated electrons B. hydrated ions C. electrons D. ions
- The current carriers that are responsible for the conductance of conductors are A. hydrated electrons B. hydrated ions C. electrons D. ions
SECTION B
- Differentiate between strong electrolyte and weak electrolyte giving examples.
- State THREE differences between electrovalent and covalent compounds.
WEEK SIX
TOPIC: ELECTROLYSIS
CONTENT
- Meaning of Electrolysis
- Definition of Terms
- Preferential Discharge of Ions during Electrolysis.
ELECTROLYSIS
Chemical decomposition of a substance can be brought about by heating. Similarly, electrical effect can also be used to bring about chemical decomposition of substances. Effect of electricity on matter is studied under electrolysis.
DEFINITION OF TERMS:
- ELECTROLYSIS: is defined as the chemical decomposition of a compound (electrolyte) brought about by the passage of direct current through either a solution or the molten form of the compound.
2. ELECTROLYTE: An electrolyte is a compound which conducts electricity and
is decomposed in the process. To behave as an electrolyte, the compound must be in a liquid form either as molten compound or an aqueous solution of the compound. A non-electrolyte does not conduct electricity in this manner.
3. ELECTRODES: Electrodes are conductors in the form of wires, rod or plates through which an electric current leaves or enters the electrolyte.
i. ANODE: Anode is the positive electrode by which the electrons leave an electrolyte (or by which conventional current enters the electrolyte). It is the electrode which is joined to the positive terminal of the direct current supply.
ii. CATHODE: This is the negative electrode by which electrons enters the electrolyte. It is the electrode which is joined to the negative terminal of the electric supply.
5. ELECTROLYTIC CELL: Electrolytic cell is an assembly of two electrodes in an electrolyte used for the electrolysis of a substance.
In an electrolytic cell, oxidation occurs at the anode (positive electrode) and reduction at the cathode (negative electrode).

EVALUATION
1. Define the following terms
[a] electrolysis [b] electrode
2. Explain the effect of electricity on matter
MECHANISM OF ELECTROLYSIS
 The electrolysis of a given electrolytes can be carried out in electrolytic cell in two forms.
The electrolysis of a given electrolytes can be carried out in electrolytic cell in two forms.
i. Using the molten form of the electrolytes
ii Using the solution form of the electrolyte
i. Using the molten (fused) form of the electrolyte, only two opposite ions form the electrolyte are present. Example, molten NaCl contains Na+ and Cl– ions only. Na+ ions migrate to the cathode to accept electrons and become discharged to produce neutral Na atoms.
 Na+(s) + e– Na(s)
Na+(s) + e– Na(s)
While chloride ions migrate to the anode to give up electrons and become discharged to produce Cl atoms which pair up to form chlorine gas, Cl2.
 Cl– Cl + e–
Cl– Cl + e–
 Cl +Cl Cl2(g)
Cl +Cl Cl2(g)
There is no competition for discharge of ions at the electrodes .
ii. Using the solution form of the electrolysis, ions are produced from the electrolytes and from the solvent usually water, H2O. Two opposite ions from the electrolyte e.g Na+ and Cl– from NaCl and two from water, H+ and OH–. In such cases, the cations and anions of both the electrolyte and the solvent will migrate to the cathode and the anode respectively where they will compete with one another to be discharged. The products formed at the electrodes depend on which ions are preferentially discharged, the ions from the electrolytes or from the solvent.
PREFERENTIAL DISCHARGE OF IONS DURING ELETROLYSIS
FACTORS AFFECTING PREFERENTIAL DISCHARGE OF IONS
The discharge of ions at the electrodes is governed by three conditions, namely
1. The position of ions in the electrochemical series.
2 The concentration of ions.
3. The nature of the electrodes
RELATIVE POSITIONS OF THE IONS IN THE ELECTROCHEMICAL SERIES

 K+
K+
Na+
Ca2+
Mg2+
Al3+
Zn2+
Fe2+
Sn2+
Pb2+
H+
Cu2+
Hg2+
Ag+
Au+


OH–
I–
Br–
Cl–
NO3–
SO42-
F–
When all other factors are constant a cation (cathode ion) which is lowest in the series (less electropositive) will be discharged in preference to the one higher up (more electropositive).This is because the less electropositive element gains electron(s) more readily from the cathode and so become reduced and discharged as a neutral atom while the more electropositive element remains in the solution as positive ions.
NOTE: K+, Na+ and Ca+ are never discharge at all from aqueous solution. This is because of the large gap between them and H+. However, K+, Na+ and Ca+ are discharged during the electrolysis of their molten salt.
An anion which is higher up in the series (less electronegative) is preferentially discharged to the one lower down the series (more electronegative). This is because the less electronegative ion loses electron(s) more readily than the more electronegative ion.
NOTE: F–, SO42- and NO3– are never discharged from aqueous solution because of the large gap between them and OH–.
CONCENTRATION OF IONS
When the concentration of an ion in the electrolyte is increased, the ion tends to increase its chances of being discharged. The influence of concentration, however, is effective only when the two competing ions are closely positioned in the electrochemical series. The effect of concentration becomes less important as the positions of the competing ions become further apart in the series.
NATURE OF ELECTRODES
Inert electrode (e.g Platinum and graphite) do not take part in the electrolytic reactions. However, platinum is attacked by liberated chlorine and graphite is attacked by liberated oxygen. Some electrodes have strong affinity (love) for certain ion and influences the discharge of such ion. For example, in the electrolysis of aqueous NaCl using mercury cathode, Na+ will be discharged at the cathode to form sodium amalgam, Na/Hg
 Na+(aq) + Hg(s) + e- Na/Hg(l)
Na+(aq) + Hg(s) + e- Na/Hg(l)
Also, in the electrolysis of CuSO4 solution using copper anode, neither the SO42- nor the OH– will be discharged. Rather, the Cu atoms will lose electrons more readily and go into solution as Cu2+ hence, the copper anode if known as reactive electrode
GENERAL EVALUATION/REVISION
1 State the modern ionic theory.
2. Name three strong and two weak electrolytes.
3. Explain how the position of a named cation and anion determine the discharge of the ion during electrolysis
4. State TWO differences between a conductor and an electrolyte
5. Describe how you can separate a mixture of lead (II) chloride, sodium chloride and ammonium chloride
READING ASSIGNMENT
New School Chemistry for Senior Secondary Schools by O. Y. Ababio (6th edition) Pages 200-204
WEEKEND ASSIGNMENT
SECTION A: Write the correct option ONLY
1. In electrolytic cell, the chemical reaction which takes place at the anode is
A. hydrolysis B. neutralization C. oxidation D. reduction
2. Which of the following is a good conductor of electricity?
A. Brine B. Ethanol C. Petrol D. Kerosene
3. Which of the statements on electrolytes and conductors is/are true?
i. Electrolytes are compounds while conductors are elements
iiElectrolytes contain mobile ions while conductors consists of mobile electrons
iii. Electrolytes can be solutions while conductors are usually solids
iv. Electrolytes and conductors can both be decomposed by an electric current
A. I, III and IV B. II, III and IV C. I, II and IIID.I, III and IV
4. Which of the following accounts for the differences in the mode of conduction of electricity by metals and aqueous solutions?
A. Electrons are present in metals but not in salt solutions.
B. Metals are conductor while salts are electrolytes.
C. Electricity is carried by mobile electrons in metals but by ions in aqueous salt solution.
D. Salts ionize in aqueous solution while metals do not.
5. Which of the following combination of ions is present in aqueous solution of sodium chloride?A. H+ and Na+ only B. H+, Na+ and OH+ only C. OH– and Cl– only D. Na+ , H+, Cl– and OH– only
SECTION B
1. Define the following terms
a. Electrolysis b. Electrolytec. Electroded. Electrolytic cell
2. Draw a diagram of a simple electrolytic cell
WEEK SEVEN
TOPIC:ELECTROLYSIS OF SOME ELECTROLTYES
ELECTROLYSIS OF CONCENTRATED SODIUM CHLORIDE SOLUTION (BRINE) USING GRAPHITE ANODE.
Ions present in solution.
NaCl H2O
Cations Na+ H+
Anions Cl– OH–
At the anode: Both Cl– and OH– migrate to the anode on passing direct current to compete for discharge. Cl– ion being more electronegative and having a high concentration in is discharged in preference to the hydroxide ion, OH–, by loss of electron to produce chlorine atom. Chlorine atoms pair to form chlorine gas.
Anodic half reaction
 Cl–(aq) Cl +e–
Cl–(aq) Cl +e–
 Cl +Cl Cl2
Cl +Cl Cl2
Overall anodic equation,
 2Cl–(aq) Cl2(g) + 2e–
2Cl–(aq) Cl2(g) + 2e–
Two moles of electrons are produced for every mole of chlorine gas formed. These are accepted at the cathode.
At the cathode: Both Na+ and H+ migrate to the cathode on passing direct current to compete for discharge. H+ being far below Na+and less electropositive is discharged by accepting electrons to produce neutral hydrogen atoms which pair up to produce hydrogen gas. Na+ concentration is high but it is very electropositive and so not discharged.
Cathodic half reaction
 H+ + e– H
H+ + e– H
 H + H H2(g)
H + H H2(g)
Overall cathode reaction
 2H+ + 2e– H2(g)
2H+ + 2e– H2(g)
Overall electrolytic reaction is obtained thus.
 2Cl–(aq) Cl2 + 2e–
2Cl–(aq) Cl2 + 2e–
 2H+ + 2e– H2(g)
2H+ + 2e– H2(g)
 2Cl–(aq) + 2H+(aq) Cl2(g) + H2(g)
2Cl–(aq) + 2H+(aq) Cl2(g) + H2(g)
The resulting solution becomes more alkaline in NaOH as concentration of Na+ and OH– increase.
EVALUATION
1. Describe the electrolysis of dilute sodium chloride solution.
2. What is the condition of the final solution?
ELECTROLYSIS OF DILUTE H2SO4 (ACIDIFIED WATER)
Acidified water is prepared by adding a few drop of tetraoxosulphate VI acid to water. It is a dilute H2SO4.
Ions present in solution
Compound H2SO4 H2O
Cations H+ H+
Anions SO42- OH–
At the anode: Both SO42- and OH– migrate to the anode. OH– is less electronegative than SO42- and is far above it in the series. It becomes preferentially discharged to produce water and oxygen gas as follows:
Anodic half reaction
 OH– (aq) OH + e–
OH– (aq) OH + e–
 OH + OH H2O + O
OH + OH H2O + O
 O + O O2(g)
O + O O2(g)
Overall anodic reaction
 4OH– 2H2O(l) + O2 + 4e–
4OH– 2H2O(l) + O2 + 4e–
At the cathode: H+ is the only cation. The ions migrate to the cathode to accept an electron each to become neutral hydrogen atoms. The atoms pair up to produce hydrogen gas molecules.
Cathodic half equation
 H+ + e– H
H+ + e– H
 H + H H2(g)
H + H H2(g)
 Overall cathodic half equation: 2H+ + 2e– H2
Overall cathodic half equation: 2H+ + 2e– H2
Overall reaction
 4OH– 2H2O(l) + O2(g) + 4e–x1
4OH– 2H2O(l) + O2(g) + 4e–x1
 2H++ 2e– H2(g) x2
2H++ 2e– H2(g) x2

 4OH–(aq) 2H2O(l) + O2 + 4e–
4OH–(aq) 2H2O(l) + O2 + 4e–
 4H++ 4e– 2H2(g)
4H++ 4e– 2H2(g)

 4OH–+ 4H+ 2H2O(l) + 2H2(g) + O2(g)
4OH–+ 4H+ 2H2O(l) + 2H2(g) + O2(g)
ELECTROLYSIS OF COPPER (II) TETRAOXOSULPHATE (VI) SOLUTION (USING DIFFERENT ELECTRODES)
Ions in solution
CuSO4 H2O
Cations Cu2+ H+
Anions SO42- OH–
At the Anode (Graphite or Platinum):Both SO42-and OH– migrate to the anode to compete for discharge. OH– is less electronegative and far above SO42-, it is preferentially discharged to produce water and oxygen.
 Anodic half reaction: OH–(aq) OH + e–
Anodic half reaction: OH–(aq) OH + e–
 OH + OH H2O(l) + O
OH + OH H2O(l) + O
 O + O O2(g)
O + O O2(g)
Overall anodic half reaction
 4OH–(aq) 2H2O(l) + O2(g) + 4e–
4OH–(aq) 2H2O(l) + O2(g) + 4e–
At the Cathode (Graphite or platinum or copper): Both Cu2+ and H+ migrate to the cathode to compete for discharge. Cu2+ is less electropositive and the concentration is high. It becomes discharged as copper atom.
 Cu2+ + 2e- Cu(s)
Cu2+ + 2e- Cu(s)
Overall cell reaction
 4OH(aq) 2H2O (l) + O2 + 4e– x1
4OH(aq) 2H2O (l) + O2 + 4e– x1

 Cu2+ + 2e– Cu(s) x2
Cu2+ + 2e– Cu(s) x2
 4OH–(aq) 2H2O (l) + O2(g) + 4e–
4OH–(aq) 2H2O (l) + O2(g) + 4e–
 2Cu2+(aq) + 4e– 2Cu(s)
2Cu2+(aq) + 4e– 2Cu(s)

 4OH–(aq) + 2Cu2+(aq) 2H2O(l) + 2Cu(s) + O2(g)
4OH–(aq) + 2Cu2+(aq) 2H2O(l) + 2Cu(s) + O2(g)
At the end of the electrolysis, the bluish colour of the electrolyte is lost and the solution becomes more acidic due to the H+ and the SO42- left in the solution.
At the Anode (Copper): Both SO42- and OH – migrate to the anode to compete for discharge. Neither SO42- nor OH– is discharged but the anode dissolves and goes into the solution as Cu2+.
 Cu (s) Cu2+ + 2e–
Cu (s) Cu2+ + 2e–
There is no change in the composition of the electrolyte since copper is merely transferred from the anode to the cathode during electrolysis.
GENERAL EVALUATION/REVISION
1. State the products formed as the electrode during the electrolysis of CuSO4solution using Cu anode
2. Write balanced half equations for the electrolysis of dilute H2SO4.
3. Describe the electrolysis of dilute sodium chloride solution and state the condition of the final solution.
4. Define the following terms: Atoms, Molecules and Ions.
5. State the Dalton’s atomic theory and its modifications.
READING ASSIGNMENT
New School Chemistry for Senior Secondary Schools by O. Y. Ababio, pages 205-207
WEEKEND ASSIGNMENT
SECTION A: Write the correct option ONLY
1. Metal X will be above metal Y in the activity series if X
A. Displaces ions of Y from solution
B. Has a higher boiling point than Y
C. Has a higher melting point than Y
D. Has a higher relative atomic mass than Y
2. What will happen at the anode when copper II tetraoxosulphate VI solution is electrolyses using platinum electrode
A. Copper is dissolved B. Copper metals is deposited
C. hydrogen is discharged D. Oxygen gas is produced
3 Oxidation takes place at the anode during electrolysis because anode
A. Is deficient in electrons B. is different in protons
C. has excess electrons D. has attraction for positive ions.
4 What product is formed at the cathode during the electrolysis of concentration sodium chloride solution using carbon electrode
A. Chlorine B. hydrogen C. oxygen D. sodium
5 Which of the following reactions occurs at the cathode during electrolysis of CuSO4 solution using copper electrodes?

 A.Cu(s) – 2e– Cu2+ (aq) B.Cu2+(aq) – 2e– Cu(s)
A.Cu(s) – 2e– Cu2+ (aq) B.Cu2+(aq) – 2e– Cu(s)

 C.Cu(s)– e– Cu2+(aq) D.Cu2+(aq) + 2e– Cu(s)
C.Cu(s)– e– Cu2+(aq) D.Cu2+(aq) + 2e– Cu(s)
SECTION B
1. State TWO differences between a conductor and an electrolyte.
2. Describe the electrolysis of CuSO4 solution using platinum electrode.
WEEK EIGHT
TOPIC: FARADAY’S LAWS OF ELECTROLYSIS
CONTENT
- Faraday First and Second laws of electrolysis.
- Verification of Faraday’s Laws of Electrolysis.
- Calculations based on Faraday’s Laws.
FARADAY’S FIRST LAW OF ELECTROLYSIS
Faraday’s first law states that the mass (m) of an element discharged during an electrolysis of an electrolyte is directly proportional to the quantity of electricity (Q) passing through it.
Faraday’s first law can be expresses in the following mathematical way
M α Q
But Q = It
M α It
M =EIt or M = EQ
Where E is the constant of proportionality known as Electrochemical equivalent of a substance.
M is measured in gram (g)
I is measured in Amperes (A)
t is measured in seconds (s)
Q is measured in Coulomb(C)
Therefore E is measured in gA-1S-1 or gC-1, for a particular electrolytic process at different time, the law can also be expressed as M1/Q1 = M2/ Q2
OR :- M1 = M2

 I1 t1 I2 t2
I1 t1 I2 t2
VERIFICATION OF FARADAY’S FIRST LAW OF ELECTROLYSIS.
The experimental set up is  The electrolytic cell above can be used to verify Faraday’s first law.
The electrolytic cell above can be used to verify Faraday’s first law.
The copper cathode is weighed at the beginning of the experiment. A steady current of 1A is passed for 10mins and the current supply switched off. The copper cathode is then weighed again to find the gain in mass. This procedure is repeated using different time intervals. A graph of gain in mass against time gives a straight line passing through the origin.
 When the experiment is repeated using different currents for a fixed period of time, a graph of gain in mass against the magnitude of the current also gives a straight line passing through the origin.
When the experiment is repeated using different currents for a fixed period of time, a graph of gain in mass against the magnitude of the current also gives a straight line passing through the origin.
 m(g) m(g)
m(g) m(g)


 t(s) I(A )
t(s) I(A )
The two graphs show that the mass of copper deposited is directly proportional the magnitude of the current, i.e M α I
EVALUATION
- State Faraday’s first law of electrolysis and write a mathematical expression for the law
- What kinds of graph are obtained when mass is plotted against time or mass is plotted against current in electrolysis?
FARADAY’S SECOND LAW OF ELECTROLYSIS
Faraday’s second law of electrolysis states that when the same quantity of electricity Q is passed through different electrolytes, the relative number of mole of the elements discharged is inversely proportional to the charges on the ions of the elements.
m = Q OR m1/Z1 = m2/Z2
Z/F M M1 M2
The minimum quantity of electricity required to liberate one mole of a univalent ion is 96500C. This quantity of electricity is called the Faraday (F).One Faraday (1F) is one mole of electron which contains Avogadro’s number (6.62 x 1023) of electrons. It follows that two Faraday’s (2F) will be required to discharged 1 mole of ions of a divalent elements or radical such as Pb2+ or S042-

 i.e M2+ + 2e- M or A2- A + 2e-
i.e M2+ + 2e- M or A2- A + 2e-
VERIFICATION OF FARADAY’S SECOND LAW OF ELECTROLYSIS
To verify the second law, set up two electrolytic cells.
i. One with copper electrodes immersed in CuSO4 solution
ii The other with silver electrodes immersed in AgNO3 solution.
The copper and silver cathodes are weighed before the experiment begins
 A steady current of 1A is passed into the solution say for 15mins and the current supply switched off. The mass of each cathode is then weighed again to find the gain in mass. The number of moles of silver deposited at the silver cathode will be doubled the number of copper deposited at the copper cathode although the same quantity of electricity was passed through the two electrolytes i.e if Y moles of copper are deposited by 2Y moles of silver are deposited. The charge on the Cu2+ is +2 and that on the Ag+ is +1 hence, the number of moles of copper and silver deposited are inversely proportional to charge on their ions
A steady current of 1A is passed into the solution say for 15mins and the current supply switched off. The mass of each cathode is then weighed again to find the gain in mass. The number of moles of silver deposited at the silver cathode will be doubled the number of copper deposited at the copper cathode although the same quantity of electricity was passed through the two electrolytes i.e if Y moles of copper are deposited by 2Y moles of silver are deposited. The charge on the Cu2+ is +2 and that on the Ag+ is +1 hence, the number of moles of copper and silver deposited are inversely proportional to charge on their ions
CALCULATIONS BASED ON THE LAW
EXAMPLES
1. Calculate the electrochemical equivalent of a metal M if a current of 5A is passed for 1hr and 2g of M is deposited
SOLUTION:
:- M = 2g, I = 5A, t = 1hr
M = Z It
2 = Z x 5x 1x 60x 60 = 2/1800
Z = 2.78 x 10-4 g I-1 s-1
2 During electrolysis of a copper solution 5g of copper was deposited in 10min when a current of 1A was passed. Calculate the time required to deposit 20g
a. if the same current was passed
b. if a current of 2A was passed.
SOLUTION:-
M1, = 5g, t1 = 10min = 600sec.
I1 = 1A
i. I1 = I2 = 1A, M2 = 200g, t2= ?
M1/ t1 = M2/t2
5/ 600 = 20/t2
t2 = 20 x 600 /5 = 2400sec.
ii I2 = 2A , M2 = 20g, t= ?
M1/I1t1 = M2/ I2 t2
5 = 20

 1 x 600 2 x t
1 x 600 2 x t
 t2 = 20 x 1 x 600
t2 = 20 x 1 x 600
2 x 5
t2 = 1200 sec.
EVALUATION
1. What mass of aluminum will be deposited if 5.6A is passed throughits solution for 10minutes?
2. Calculate the mass of silver deposited when a current of 2.6A is passed through a solution of a silver salt for 70 minutes. (Ag = 108; 1 Faraday = 96500C)
WORKED EXAMPLES
1. How many moles of electrons are required to discharge 2.5 moles of silver ion during electrolysis?
SOLUTION:
 Ag++ e– Ag
Ag++ e– Ag
1 Mole Ag+ is discharged by 1 mole e–
2.5 moles Ag+ are discharged by 2.5 mole e–
2.5 moles of electrons are required
2. If 6 moles of electrons are passed through different solutions of copper and silver, calculate the moles of silver deposited by the time 3 moles of copper have been deposited
SOLUTION
 Cu2+ + 2e– Cu
Cu2+ + 2e– Cu
1 mole of Cu is deposited by 2 moles of e–
3 moles Cu is deposited by 6 moles of e–
For silver:
 Ag+ + e– Ag
Ag+ + e– Ag
1 mole of e– deposits 1 moles of Ag
6 moles of e–will deposit 6 moles of Ag
EVALUATION
- How many Faraday’s of a quantity electricity are required to produce 2 moles of oxygen gas during electrolysis?
- A current of 0.25A flows for 16 minutes 5 seconds and deposits 0.163g of a metal X during the electrolysis of an aqueous solution of a salt of the metal. What is the charge on the metal ion?
APPLICATIONS OF ELECTORLYSIS
Electrolysis is applied in the industries in the following areas e.g
1. Extraction of elements: Na, K, Mg, Ca, Al, Zn and non metale.g H2, F2, Cl2 are obtained either by electrolysis of their ores or by the electrolysis of their fused compounds or their aqueous solutions.
2. Purification of metals e.g Cu, Hg, Ag, Au
3. Electroplating of one metal by another
4. Preparation of certain important compounds such as sodium hydroxide, and sodium trioxochlorate(V)
EXTRACTION AND PURIFICATION
Very electropositive metalse.gCa, Mg, and Al and electronegative non-metal e.g F and Cl are extracted from their compounds by electrolysis.
The less electropositive metalse.g copper, mercury, silver and gold occur in the uncombined ores mixed with impurities. The pure metals are obtained from the ore by electrolytic purification.
During electrolytic purification of copper ores, the impure copper is made the anode while a pure copper is made the cathode. The electrolyte is usually copper (II)tetraoxosulphate(VI) solution.
At the anode :- The metallic copper atoms in the ore give up two electrons each forming copper II ions and move into the solution.
 Cu(s) Cu2+(aq) + 2e–
Cu(s) Cu2+(aq) + 2e–
Electrolytic Purification of Copper
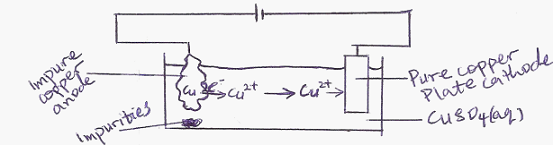
At the Cathode. The copper ions in the solution are discharged at the cathode by receiving electrons to become neutral solid copper.
 Cu2+(aq) + 2e– Cu(s)
Cu2+(aq) + 2e– Cu(s)
The net result is that pure copper atoms are transferred from impure ore (anode) to the cathode. The impurities are collected at the base of the cell used.
EVALUATION
- Mention two metal extracted by electrolysis
- Give two industrial application of electrolysis
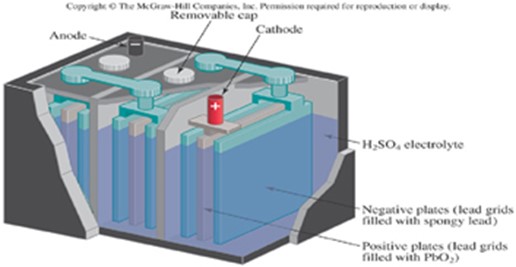
ELECTROPLATING
Electroplating is a method of coating the surface of one metal with another metal, usually copper, silver, chromium, nickel, or gold by means of electrolysis.
The main objectives of electroplating are
1. Decoration
2. Protection against corrosion.
Article which are usually plated include cutlery, jewellery and iron or steel objects such as the metallic parts of motor cars
Silver – plating of a spoon
In Silver plating of an iron spoon
a. The anode is a silver rod
b. The cathode is the spoon
c. The electrolyte is a soluble silver salt, usually a silver trioxonitrate V solution
 At the anode: Ag(s) Ag+ + e–
At the anode: Ag(s) Ag+ + e–
 At the cathode: Ag+(aq) + e– Ag(s)
At the cathode: Ag+(aq) + e– Ag(s)
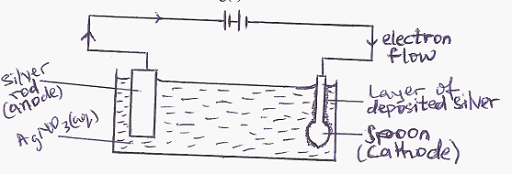
GENERAL EVALUATION/REVISION
- In silver – plating an iron spoon, which electrode is the spoon made?
- Electrolytes used in silver – plating is.
- Mention two metal extracted by electrolysis
- Give two industrial application of electrolysis
- Briefly describe purification of impure copper ore.
READING ASSIGNMENT
New School Chemistry for Senior Secondary Schools by O. Y. Ababio (6th edition) pages 210-212.
WEEKEND ASSIGNMENT
SECTION A: Write the correct option ONLY
1 In silver plating a spoon, the spoon must be made the
a. Anode b. Cathode c. Electrode d. Electrolyte
2 A current of 20A is passed for 30 minutes during the electrolysis of gold (III) tetraoxosulphate (VI). The mass of gold deposited at the cathode is
a. 24.4g b. 26.5g c. 28.9g d. 32.6g
3 What quantity of chlorine in moles is liberated when a current of 15A is passed through molten potassium chloride in 1930 seconds?
a. 0.05 b. 0.10 c. 0.15 d. 0.20
4 What quantity of silver is deposited when 96500C of electricity is passed through a solution containing silver ions [Ag = 108, F = 96500C]
a. 1.08g b. 10.8g c. 108g d. 5.40g
5 Calculate the volume of oxygen liberated at s.t.p when 96500C of electricity is passed through aqueous solution of H2S04. [IF = 96500C, Volume at s. t. p = 22.4 dm3]
a. 5.6dm3 b. 0.56dm3 c. 56dm3 d. 560dm3
SECTION B
1. In the electrolysis of dilute CuSO4 solution using platinum electrodes, 2.0A current passed for 2.50hrs and deposited 5.85g of copper
a. Calculate the relative atomic mass of copper from the experiments
b. What volume of gas is liberated at s.t.p ?
2. a. Using a well-labeled diagram only, explain the process of silver plating a spoon.
b. Give three uses of electrolysis
WEEK NINE
TOPIC: ELECTROCHEMICAL CELLS
CONTENT
- Electrolytic and Electrochemical Cells with their differences.
- Drawing and Writing of Cell Diagrams.
- Standard Electrode Potential.
- Calculation of e.m.f of a Cell.
ELECTROLYTIC AND ELECTROCHEMICAL CELL
Electrolytic cell is a device in which electrical energy is converted to chemical energy. An electrochemical cell is a device in which chemical energy is converted to electrical energy.
DIFFERENCES BETWEEN ELECTROLYTIC CELL AND ELECTROCHEMICAL CELL
ELECTROLYTIC CELL ELECTROCHEMICAL CELL
1. Electrical energy is converted to Chemical energy is converted energy
chemicalenergy. electrical energy.
2. Cathode is negative electrode while anode Cathode is positive electrode while
is positive electrode. anode is negative electrode.
3. No salt bridge is required Salt bridge or porous partition required. is required.
4. Electrolyte may not contain ions of Electrolyte must contain metal ions of metal electrode used.electrode used
5. Electrons are pushed by an external source Electrons are generated by oxidation such as battery. at the anode.
Electrolytic cell is also known as galvanic cell it consists of two half cells
1. an oxidation half cell
2. a complementary reduction half-cell.
DRAWING AND WRITING OF CELL DIAGRAMS
A typical electrochemical cell consists of two electrodes (cathode and anode) and their electrolytes. The electrodes are connected by means of a salt bridge. A typical electrode or half-cell is a device in which an element (metal) is in contact with its own ions. A typical metallic electrode is symbolically represented as:
M(s)/Mn+ (aq) or Mn+ (aq)/M(s)for metals dipped into solutions of their own ions.
Conventional notation is used to describe an electrochemical cell without drawing. Example:
X/Xn+ //Yn+/Y
The single vertical lines (/) represents metal and metal ion interphase while the double lines (//) represents the salt bridge or a porous partition.
For a simple electrochemical cell, there is flow of electron from the anode (negative electrode) to the cathode (positive electrode). Also, for an electrochemical to produce electricity, the anode element must be more reactive than the cathode element. That is, the anode element must be higher in the electrochemical series than the cathode.
For a given net reaction as shown below
 Zn(s) + Cu 2+(aq) Zn2+(aq) + Cu(s)
Zn(s) + Cu 2+(aq) Zn2+(aq) + Cu(s)
The Cell notation for the system is
Zn/Zn2+ // Cu2+/ Cu
Since Zn is higher up in the electrochemical series, it will serve as the anode while copper will serve as the cathode. The anodic half-cell is always written at the left hand side while the cathodic half-cell is always the right hand side.
The diagram below shows the drawing of the above electrochemical cell:

Salt Bridge
In an electrochemical cell, instead of using the porous partition to maintain the electrical neutrality of the half-cell electrolytes, the half-cells can be linked by a SALT BRIDGE. Salt bridge is a filter paper soaked in sodium chloride solution or KCl or NH4NO3.
Functions of a salt bridge
1. It helps in completion of electric circuit
2. It enables the migration of ions from one half cell to the other in order to maintain electrical neutrality in the solution when current flows in the set up
EVALUATION
- State two differences between electrolytic and electrochemical cells.
2a. Write a cell notation for the equation below
 Zn(s) + 2Ag+(aq) Zn2+(aq) + Ag(s)
Zn(s) + 2Ag+(aq) Zn2+(aq) + Ag(s)
- Draw the cell diagram for the cell above.
ELECTRODE POTENTIAL
This is the potential difference that is set up between metallic electrode and its electrolyte. Example
i. Copper ion/copper system:
If a copper plate is dipped into a CuSO4 solution, the net reaction is
 Cu2+(aq) + 2e– Cu(s)
Cu2+(aq) + 2e– Cu(s)
That is: Cu2+(aq)/Cu(s)
ii. Zinc ion/zinc system:
If a zinc plate is dipped into a ZnSO4 solution; the net reaction is
 Zn2+(aq) + 2e– Zn(s)
Zn2+(aq) + 2e– Zn(s)
That is: Zn2+(aq)/Zn(s)
Standard Electrode Potential
The electrode potential of a given system depends on
- the overall energy change
- the concentration of ions in the solution
- the temperature
Standard electrode potential (E0) is used to compare the electrode potential values of different metal ions/metal system.
In measuring standard electrode potential of metal ion/metal system difference (in volts) which exists between the metallic electrode and the standard hydrogen electrode, the standard hydrogen electrode potential is considered to have an arbitrary value of zero.
The standard electrode potentials of a metal ions/metal system is the potential difference set up between the metal and its ion compared to electrode potential of H+(aq)and H2 gas system.
Metal ion/Metal E0
K+(aq)/K(s) -2.92V
Ca2+(aq)/Ca(s) -2.87V
Na+(aq)/Na(s) -2.71V
Zn2+(aq)/Zn(s) -0.76V
2H+(aq)/H2(g) 0.00
Cu2+(aq)/Cu(s) +0.36V
Ag+(aq)/Ag(s) +0.80V
Electrode potential of Cu = +0.34V as electron flows from the hydrogen electrode to copper electrode. At platinum hydrogen electrode
 H2(g) 2H+ + 2e–(oxidation)
H2(g) 2H+ + 2e–(oxidation)
At the copper electrode
 Cu2+(aq) + 2e– Cu(s) (reduction)
Cu2+(aq) + 2e– Cu(s) (reduction)
 Overall reaction: Cu2+(aq)+ H2(g) Cu(s) + 2H+(aq)
Overall reaction: Cu2+(aq)+ H2(g) Cu(s) + 2H+(aq)
NOTE: Eǿtotal is negative if electron flows from the metal electrode and positive when it flows from hydrogen electrode to the metal electrode
EVALUATION
- Define electrode potential
- List two functions of a salt bridge.
E.M.F OF A CELL
When two half-cells are joined together through a salt-bridge, thee.m.f of the cell thus formed is the algebraic difference between the two electrode potentials.
The e.m.f. of the cell formed by the system Zn(s)/Zn2+(aq)//Cu2+(aq)/Cu(s) is defined as the standard electrode potential of the right hand electrode minus the standard electrode potential of the left-hand electrode.
Eǿtotal = EǿR– EǿL
= +0.34 – (-0.76)
=+1.10V
A positive e.m.f. indicates that the left- hand electrode(zinc) is partially capable of reducing copper (ii) ions to copper
But for the system Cu(s)/Cu2+(aq)//Zn2+(aq)/Zn(s)
Eǿtotal = EǿR – EǿL= -0.76-0.34= -1.10V
A negative e.m.findicates that the left-hand electrode (copper) cannot reduce zinc ions to zinc atom.
In general, when this convention is used, then a positive e.m.f indicates that the reaction is thermodynamically feasible as written down from left to right.
A negative e.m.f implies that the reaction is thermodynamically impossible as written down.
GENERAL EVALUATION/REVISION
1. Calculate the e.m.f of a cell when zinc half cell is connected with a salt bridge to a copper half cell if the Eө of Zn2+(aq)/Zn(s) = -0.76V and Ag+(aq)/Ag(s) = +0.80V
2. Give two examples of primary cells
3. Define electrode potential and list two functions of a salt bridge.
4. What is standard electrode potential?
5. State Aufbau principle and Hund’s rule of maximum multiplicity.
READING ASSIGNMENT
New School Chemistry for Senior Secondary Schools by O. Y. Ababio, pages 215-216
WEEKEND ASSIGNMENT
SECTION A: Write the correct option ONLY
1. A Porous materials used to maintain electrical neutrality in solution of an electrochemical cell is called A. Pot B. Salt bridge C. Paper D. none of the above
2. The element with the electronic configuration 1s22s22p63s23p64s2 is a/an
A. group two element B. transition metal C. alkaline earth metal D. none of the above
3. Oxidation takes place at the anode during electrolysis because anode
A. is deficient in electrons B. is different in protons C. has excess electrons D. none of the above
4. The potential difference set up between metallic electrode and the electrolyte solution is known as A. Lead accumulator B. electrode potential C. Voltammeter
D. none of the above
5. In an electrochemical cell, the cathode is made A. negative B. positive c. neutral
d. none of the above
SECTION B
1a. State THREE differences between an electrolytic cell and an electrochemical cell
b. Mention TWO function of a salt bridge
2. What is the voltage of the cell represented as Ca(s)/Ca2+(aq)//Cu2+(aq)/Cu(s) given that electrode potential of Ca2+(aq)/Ca(s) is -2.87V and that of Cu2+(aq)/Cu(s) is +0.36V
WEEK TEN
TOPIC:APPLICATION OF ELECTROCHEMICAL CELLS
CONTENT
- Distinction between Primary and Secondary Cells
- Types of Electrochemical Cells.
APPLICATION OF ELECTROCHEMICAL CELLS
Electrochemical cells are widely used to generate small currents of electricity for everyday purposes and industrial operations.
PRIMARY AND SECONDARY CELLS
Electrochemical cell can be classified into: primary cell and secondary cell.A primary cell is that is not rechargeable e.g Daniel cell and Leclanche cell. A secondary cell is a cell that is rechargeable e.g Lead acid accumulator.
The distinction between a primary and a secondary cell is as follows:
- A primary cell cannot be recharged whereas a secondary cell can be recharged.
- In a primary cell, chemical reaction is irreversible whereas in a secondary cell, chemical reaction is reversible.
- A primary cell is cheap and easy to use while a secondary cell is expensive and complex in usage.
Primary cells are commonly found and used in most of the electric toys, clocks, wrist watches, domestic remote control, etc.
Secondary cells are commonly used in starting, lighting and ignition in vehicles. They are also in Uninterrupted Power Supplies (UPS), telecommunications, etc.
TYPES OF ELECTROCHEMICAL CELLS
- DANIEL CELL: It consist of copper vessel filled with saturated solution of copper (II) tetraoxosulphate (VI) and a porous pot filled with a zinc tetraoxosulphate (VI) solution where a zinc rod is immersed in. The initial e.m.f produced by the Daniel cell is about 1.1volts. This cell has to be replaced once the chemicals in it are used up.
- LECLANCHE CELL: For this cell, the anode and cathodeare a zinc rod and carbon rod respectively. The electrolyte is an ammonium chloride in solution or paste form.
At the anode, the zinc atoms lose two electrons each to dissolve in the electrolyte as positively charged zinc ions. The electrons which are released pas through an external circuit performing useful work such as lighting-up a torch bulb before arriving at the cathode of the cell.
Zn(s)→ Zn2+(aq) + 2e– Oxidation
At the cathode, the ammonium ions accept electrons to become reduced to ammonia and hydrogen gas. The hydrogen gas is removed by manganese (IV) oxide to prevent it from adhering to the cathode. This is known as depolarization of the cell.
2NH4+(aq) + 2e–→ 2NH3(g) + H2(g) Reduction
The Leclanche dry cell is the most common cell used in many small electrical appliances such as torches, transistor radio, bicycle lamps etc.
- LEAD ACID ACCUMULATOR: This commonly referred to as car battery. It is often charged by passing direct current through it. The charged cell can then produce an electric current when required. The cathode of a fully-charged lead accumulator is lead (IV) oxide, PbO2, and its anode is metallic lead. The electrolyte is dilute tetraoxosulphate (VI) acid solution.

For a discharging cell:
At the anode: The lead atoms release two electrons each to become oxidized to lead (II) ions which then combine with tetraoxosulphate (IV) ions in the electrolyte to become deposited on the anode as lead (II) tetraoxosulphate (VI). The electrons released pass round an external circuit as an electric current to be used for starting the engine of a vehicle, for lighting the car lamps and so on before arriving at the electrode.
Pb(s)→ Pb2+(aq) + 2e–
Pb2+(aq) + SO42-(aq)→ PbSO4(s)
At the cathode: The electrons from the anode are accepted at the cathode where the lead (IV) oxide and the hydrogen ions from the electrolyte undergo reduction to produce lead (II) ions and water. The lead (II) ions then combine with the tetraoxosulphate (VI) ions from the electrolyte to become deposited at the cathode as lead (II) tetraoxosulphate (VI).
PbO2(s) + 4H+(aq) + 2e–→ Pb2+(aq) + 2H2O(l)
Pb2+(aq) + SO42-(aq)→ PbSO4(s)
During the discharging process, the density of the acid decreases to 1.15gcm-3 due to the absorption of hydrogen and tetraoxosulphate (VI) ions from the electrolyte and the e.m.f of the cell drops to 1.8V.
When both electrodes are completely covered with lead (II) tetraoxosulphate (VI) deposits, the lead accumulator will stop discharging a current. To make produce a current again, it will have to be recharged.
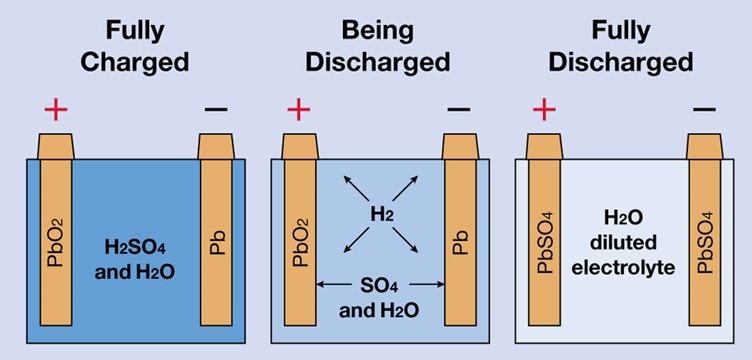
For a recharging cell: The electrode which is the anode in the charged cell becomes the cathode, while the cathode becomes the anode. The anode and cathode of an external electrical source are connected to the cathode and anode of the recharging cell respectively. The reactions are summarized as follows:
At the cathode:
Pb2+(aq)→Pb(s)
PbSO4(s)
SO42-(aq)
At the anode:
Pb2+(aq)→ PbO2(s)+ 4H+(aq)
PbSO4(s)
SO42-(aq)
After recharging, the density of the acid in the cell returns to the initial value of 1.25gcm-3 due to the release of the same amount of ions (4H+, 2SO42-) into the solution and the e.m.f of the cell returns to 2.2V.
GENERAL EVALUATION/REVISION
- Describe the structure of the Leclanche cell.
- Describe the chemical reactions that take place inside a lead acid accumulator.
- State the function of manganese (IV) oxide in a Leclanche cell.
- State Faraday’s second law of electrolysis.
- Define the electrode potential of a metal.
READING ASSIGNMENT: New School chemistry for Senior Secondary School by O. Y. Ababio (6th edition) Pages 217-218.
WEEKEND ASSIGNMENT
SECTION A: Write the correct option ONLY
- An electrolytic cell uses electrical energy to drive A. chemical reaction B. physical reaction C. no reaction D. none of above
- An electrochemical cell is also called A. battery cell B. galvanic cell C. cell D. chargeable cell
- Electrochemical cells convert A. mechanical energy in to electrical energy B. potential energy in to electrical energy C. kinetic energy in to electrical energy D. chemical energy in to electrical energy
- In the activity series of metals, metals higher up will serve as A. cathode B. anode C. salt bridge D. none of the above.
- Electrolyte of dry Leclanche cell is A. anhydrous ZnSO4 B. pasty MnO2 C. NH4Cl paste D. muslin bag.
SECTION B
- Describe the structure of the Leclanche cell and give the chemical reactions that take place in it.
- The reactions taking place inside a discharging lead acid accumulator are said to be redox reactions. Give these chemical reactions and justify the statement.