MATTER
CONCEPT OF MATTER
MATTER
Is anything that occupies space and has weight. Including all common materials around animals.
- Solid- For example: stones, pen, paper, nails, door, etc.
- Liquid- For example: water, juice, alcohol, tea etc.
- Gas- For example: oxygen, (O2), nitrogen (N2) etc.
The kinetic nature of matter to explain the existence of matter in the three states solid ,liquid and gas.
Kinetic nature of matter
IN SOLID
– Solid have a definite shape .They do not conform to the shape of the container. Also they have definite volume.
IN LIQUID
The particles of substance have more kinetic energy than those in a solid .The liquid particles are not held in a regular arrangement, but are still very close to each other. So liquids have a definite volume -liquid like solid ,can not be compressed .Particles of liquid have just enough room to flow around each other . So liquids have an indefinite shape.
IN GASES
Gas particles have a great deal of space between them and have high kinetic energy .If unconfined, the particles of a gas will spread out indefinitely.If confined, the gas will expand to fill its container.
Relationship between the states of matter
4. Condensation- Is the changing of gas to liquid.
(importance of changing one state of matter to another)
A simple example of matter .If you have a block of ice, you have solid water .Add heat (a form of energy ) and the ice melts into liquid water that you could drink ( it has reached its melting point) continue to apply heat, and the water will evaporate and turn to steam which is water in a gaseous state (it has reached boiling point). This works backward too, gas can cool down by losing energy and condense back into liquid water and cool down farther into a solid. This process is called sublimation where a solid can turn straight into a gas when heat is applied
- Formation of rainfall i.e. condensation.
- To help us make business.
- To help us in the separation of mixtures, eg. separating sugar and H2O
- Production of salt.
- Burning of water to form vapor
- Melting of solid to liquid example ice to water
- Magnetization of iron
- Grinding of chalk
- Breaking of glass into smaller pieces
- Condensation of steam into water
- Dissolving salt into water
- Dissolving sugar in water
- Do not produce new kind of matter
- It is generally reversible
- No change in mass
- No heat is applied
- Burning of paper
- Decaying of teeth
- Souring of milk
- Rusting of iron
- Decaying of food
- New kind of matter is formed
- The change is irreversible
- There is change in mass
- It involves heat change
- Metals
- Non metals
- They are good conductors of heat and electricity.
- They have high melting and boiling points
- They are strong and tough
- They are bright and shiny
- They have high density
- They have molecules
- They make noise when you bang them.
- Silver
- Potassium
- Sodium
- Magnesium
- Aluminum
- Lithium
- Silicon
- Iron
- Chlorine
- Fluorine
- Bromine
- Iodine
- Oxygen
- Sulphur
- Phosphorus
- They are bad conductors of heat and electricity
- They have low melting and boiling points
- They have low density
- They are not malleable
- They are not bright
- They are not strong and tough
- They are not shiny
METALS | NON METALS |
They are good conductors of heat and electricity | They are bad conductors of heat and electricity |
They have high melting and boiling points | They have low melting and boiling points |
They are strong and tough | They are not strong and tough |
They are bright | They are not bright |
Have high density | They have low density |
They are malleable | They are not malleable |
Sonority | They do not have sonority |
Chemical symbol
OR.
Name and symbols to chemical elements
Assign name and symbols to chemical elements .
(i) An element may be represented that is derived from the first letter of English name
Examples.
NAME SYMBOL
Carbon C
Oxygen O
Phosphorus P
Sulphur S
Hydrogen H
Iodine I
Nitrogen N
Fluorine F
(ii) Names of different elements may have the same first letter for example calcium and copper. It thus necessary to differentiate the element .In this case another letter usually the second from the name is used with the first one .The first letter will be capital while the second will be a small letter (not capital)
Examples.
NAME SYMBOL
Calcium Ca
Chlorine Cl
Cobalt Co
Magnesium Mg
Manganese Mn
Aluminum Al
Argon Ar
(iii) In some cases ,the symbols are derived from Latin names instead of the common English names.
Examples.
NAME LATIN
Sodium Nautrium
Potassium Kalium
Copper Cuprum
Iron Ferrum
Mercury Hydrargyrum
Silver Argentum
Gold Ausum
Tin Stannum
Lead Plumbum
Element | Symbols | Elements | symbols |
Sodium | Na | oxygen | O |
potassium | K | chlorine | Cl |
magnesium | Mg | Fluorine | F |
copper | Cu | Neon | Ne |
lithium | Li | Argon | Ar |
silicon | Si | Helium | He |
Iron | Fe | Nitrogen | N |
Gold | Au | Barium | Ba |
sulphur | S | Barron | Br |
Aluminum | Al | phosphorous | P |
Silver | Ag | Lead | Pb |
Zinc | Zn | Carbon | C |
COMPOUND AND MIXTURES
- Water( H2O)
- Salt (NaCl)
- Sodium carbonate (Na2CO3 )
- They cannot be separated by physical means.
- The properties of compounds are different from the ones of an element.
- Heat is usually given out or absorbed with a compound.
i. Oil and water
ii. Blood
ii
i. Air
iv. Urine
v. Ink
- Homogeneous mixture- is a mixture which has uniform composition, appearance and properties.
- Heterogeneous mixture- is a mixture which has non-uniform compositions, appearance and properties.
Mixture | compounds |
Components may be seen separately | Components cannot be seen separately |
The components can be separated by physical means | The components can be separated by chemical means |
The elements are mixed in any proportions | The element proportion is fixed |
No chemical change takes place when it is formed | Chemical change takes place in the substance formed. |
The properties are those of the individual elements | The properties are different from those of individuals. |
Solvent – Is a substance that dissolves the solute. E.g. water
- It is applied in the separation of mixtures in the laboratory
- Extraction of some mineral such as common salt
- Decantation
- Evaporation
- Distillation (simple distillation and fraction distillation)
- Sublimation
- Filtration
- Chromatography
- Layer separation
- Solvent extraction.
Is the process of separating a solute from a liquid.
Is the process of separating a mixture by heating a liquid to a very high temperature until it vaporizes in other words turns into a gas.
(a) Simple distillation
(b) Fractional distillation
7.Chromatography
1.Define the following methods of separating mixtures
- Sublimation – is the process whereby a solid changes state directly to gas usually on heating.
- Decantation – is the process of separating a heterogeneous mixture of a liquid and a solid.
- Filtration – is the method used to separate a heterogeneous mixture of a solid and a liquid.
- Distillation – refers to the process of separating liquids to a very high temperature until it vaporizes burns into gas.
- Layer separation – is the method used for separating immiscible liquids using a separating funnel.
- Used and applied in softening hard water
- Applied in obtaining cooking oil from plants and animal
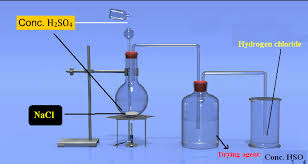
4. Draw and label the diagram of fractional distillation
REVIEW QUESTION
1. (a) Explain the meaning of matter.
(b) Describe each of the three state of matter.
(c) Explain importance of changing one state of matter to another.
2. Define a/ (i) chemical change.
(ii) physical change.
b/Give the characteristics of each of the above.
c/ Give the differences between physical change and chemical change.
3.Define a/ (i) Metal.
(ii) Non metals.
b/ Different between metal and non metal.
4. Define the following a/ (i) compound
(ii) mixture
b/ Give the differences between compound and mixtures.
5. Define the following
a/ (i) solutions.
(ii)suspensions.
(iii) emulsion.
b/ mention three classes of mixture.
6. Define the following
(i) Sublimation .
(ii) Decantation.
(iii) Filtration.
(iv) Distillation.
(v) Layer separation.





6 Comments