LEARNING OBJECTIVES
By the end of this chapter, you should be able to:
1. (a) Define: Expansion and Contraction of a material.
(b) Describe: The ball & ring / The bar & the gauge experiments to demonstrate expansion and contraction in solids.
(c) State (i) The applications of expansion in:
(ii) The problems caused by expansion and their solutions.
2. (a) Define: Linear expansivity
(b) Solve problems involving linear expansivity.
3. (a) Describe: Experiments to show expansion in Liquids and Gases.
(b) Explain the anomalous behaviour of water and give its importance to aquatic animals.
4. (a) State: (i) Any two thermometric liquids.
(ii) The properties of thermometric liquid.
- Advantages of mercury over alcohol as used in a thermometer.
- Define: The lower and upper fixed points.
- Solve problems involving
(i) Conversion of temperature from one scale to another.
(ii) Calculation of temperature for unmarked thermometer.
15.1 Expansion and Contraction
Expansion is the increase in size of an object when it gets hotter, while contraction is the decrease in size of an object when it becomes colder.
(a) Expansion of solids
All solids expand when heat is applied to them. However, they do so in varying amounts. Some solids expand very little while others expand greatly.
Expansion in solids can be investigated by using the following experiments:
- The ball and ring.
- The bar and gauge.
Experiments to investigate the effect of heat on solids
Experiment 15.1 Ball and ring experiment
Apparatus/Requirements: A ball and ring, source of heat.
Procedure Part I:
Pass the ball through the ring when cool as shown in figure 15.1 below.
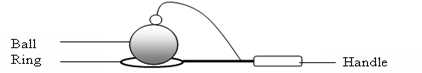
Figure 15.1
Observation: The ball passes through the ring easily.
Part II:
- Heat the ball on a Bunsen burner for some time.
- Try to pass it through the hole again.
Observation: The ball does not pass through.
Explanation: When the ball is heated, it expands, i.e., the size becomes bigger. As such, it cannot pass through the ring.
Experiment 15.2 The bar and gauge experiment
Apparatus/Requirements: A bar and gauge, source of heat.
Procedure Part I:
- Pass one end of the bar through the hole on the gauge.
- Remove the bar and fix it between the ends of the gauge as shown in figures 15.2 (a) and (b).

Observation: The head of the bar passes through the hole and also fits in the gauge easily.
Part II:
- Heat the bar on a Bunsen burner flame for some time.
- Try to pass it through the hole and also try to fix it in the gauge.
Observation: The head of the bar does not pass through the hole and does not fit in the gauge.
Allow the bar and the ball to cool and repeat the above procedures of passing the ball through the ring and fixing the bar in the hole and the gauge respectively.
Results: When the ball and the bar cool:
- The ball passes easily.
- The bar fits in the gauge easily.
Explanation
When the bar is heated, it expands both sideways and lengthwise (i.e., the size becomes bigger and longer). As such, it cannot pass through the hole and fit in the gauge.
Note: The gauge shows that the bar has increased in length, which is called linear expansion. The ring shows that the diameter of the ball has increased in all directions. The expansion in area of a solid is known as superficial expansion, and the expansion in volume is called cubical expansion.
15.11 Applications of expansion in solids
(a) Bimetallic strip A bimetallic strip is a strip made of two different metals welded or riveted together. When cold, the double strip is straight as shown in figure 15.3 below.

(b) Effect of heat on bimetallic strip
When a bimetallic strip is heated, the metals expand with the metal of higher expansivity expanding more than the one of lower expansivity. The strip bends to form a curve with the metal of high expansivity on the outside as shown in figure 15.4 below.

Examples of a bimetallic strip are:
Brass and iron – Brass having higher expansivity than iron.
Aluminium and iron – Aluminium having higher expansivity than iron.
Question: State which metal would be on the outside and inside if the two bimetallic strips in the example above are heated.
(c) Uses of expansion in bimetallic strip
Bimetallic strips are used as electrical switches, in thermostats, and many other mechanical switching devices.
Thermostat
A thermostat is a device that automatically regulates the temperature of a system by maintaining it constant or varying it over a specific range.
A bimetal thermostat uses a special strip of metal to open and close a circuit as temperature fluctuates. Two metals with different expansion rates are bonded to make the strip. The thermostat is arranged so that when the metals are hot, the strip bends upward (toward the metal with the lower expansion rate) and disconnects the circuit. In this particular case, the thermostat will activate a heater when the circuit is closed and electricity is flowing.

Note: Increasingly, the use of bimetallic strips for this purpose is being replaced by electronic circuits with no moving parts.
(a) Tight fitting of
(i) Riveting
Expansion and contraction is used in riveting to get a tight joint of two or more metal plates.
- A hot rivet (expanded) is pushed through a hole in the two plates or rods to be joined.
- The end of the hot rivet is hammered to form another.

Results: As the rivet cools, it contracts and pulls the two plates more tightly.
(ii) Steel tyres onto cart and train wheels
The steel tyre is designed to just fit when it is red hot. As it cools down, it tightens and grips on the wheel.
(iii) Wheel and axles
The same method is also used in fitting wheels onto axles.
(b) Steel bridges
Girder bridges made of steel contract and become shorter during cold weather, and expand and become longer during hot weather. To allow for expansion and contraction, one end of the bridge is fixed and the other end is placed on rollers as shown in figure 15.6 below. This enhances the to-and-fro movement during expansion and contraction.

(c) Railway lines
Railway tracks have been bent and seriously damaged due to expansion during very hot days where the gap allowed for expansion was too small. Now, to allow room for expansion and contraction, fairly large gaps are left between the sections of rails. The sections are held together by fish plates fixed by bolts in oval-shaped holes.
(d) Pipelines
Pipelines carry steam, liquids, and gases from one point to another. The pipes contract when cold and expand when hot. To avoid breakage of the pipes due to the force of expansion and contraction, the pipes are fitted with loops or expansion joints. The joints and loops allow the pipes to expand and contract when steam passes through and when it cools down.
(e) Electricity and telephone lines
During cold weather, especially at night, telephone wires contract, and when it is hot during the day, they expand. During cold weather (from evening up to early morning hours), electricity/telephone wires contract. The wires become shorter and taut. During hot afternoon hours, the wires become longer and slack.
To avoid the wires from breaking, they are fixed loosely to allow contraction and expansion.
15.2 Linear expansivity
The change in the length of a substance during expansion is called linear expansion. The measure of the tendency of a particular material to expand is called its expansivity.
The lengthwise expansivity of a material is called its linear expansivity and is given by the symbol α (alpha).
Formula of linear expansivity
Definition: Linear expansivity is defined as: The increase in length of a unit length of a material for a degree rise in temperature.
i.e., Linear Expansivity = 
S.I Unit of Linear expansivity
The S.I unit of linear expansivity is a derived unit.
= 
= °C-1 or K-1
By rearranging the formula for linear expansion, we obtain a formula which can be used to calculate expansion of things like bridges and railway lines.
Linear expansion = Linear expansivity × Original length × temperature rise
Factors which determine linear expansivity
The linear expansion or change in length Δl of a material depends on three things:
(a) The length of the material, l.
(b) The change (rise) in temperature.
(c) The linear expansivity of the material.
Worked example
1. Calculate the linear expansion of an aluminium cable 50 m between two electric poles when the temperature rises by 40°C. The linear expansivity of aluminium is 2.6 × 10-5/°C.
Solution:
Δl = ?
α = 2.6 × 10-5/°C, l = 50 m, Δθ = 40 °C
Δl = α × l × Δθ
= 2.6 × 10-5 × 50 m × 40 °C
= 5.2 × 10-2 m
Answer: The aluminium will increase in length by 5.2 × 10-2 m or 0.052 m or 5.2 cm.
15.3 Expansion in liquids
All liquids, like solids, expand with varying amounts. Some expand more than others.
Experiment 15.3 To demonstrate expansion of water
Apparatus: A round bottom flask, capillary tube, source of heat, a cork, and a liquid.
Procedure
- Fill a round bottom flask up to the brim with liquid (e.g., water).
- Insert a capillary tube through a cork and cork the flask.
- Mark the level of liquid in the glass tube.
- Set up the apparatus as shown in figure 15.7 below.

Heat the flask as you observe the level of the water in the tube.
Observation: First the level of the water falls and then starts to rise again.
Explanation
(i) The fall of water level
The initial fall of water level is due to the expansion of the glass flask, which gets heated first and expands. The expansion of the flask results in increased volume of the flask. So water moves in to fill the extra volume.
(ii) The rise of water level
Finally, the heat reaches the water and starts to expand, thus rising up the glass tube.
Note that: Water expands faster than glass.
15.31 Comparison of expansion of different liquids
Expansion in different liquids can be compared by filling the liquids in different flasks of the same size and type.
Experiment 15.4 To compare the expansion of different liquids e.g., water, alcohol, methylated spirit, ether, benzene
Apparatus: Four identical flasks, trough, water, stirrer, source of heat.
Procedure
- Fill the flasks with the liquids to the same height / level and then place them in water in a trough as shown in figure 15.8.
- Heat the water bath while stirring.
- Observe the levels of the liquids in the flasks.

Observation: The levels of the liquids in the tubes first fall and then rise by different amounts as the heating continues.
Result: The result shows that ether expands most, followed by benzene, while water expands the least.
Conclusion: The above observation shows that some liquids expand more than others for a given rise in temperature.
15.32 The expansion of water
Most liquids contract steadily as they cool, and contract further on reaching their freezing point. Water contracts as it cools down from 100°C to 4°C. However, between 4°C and 0°C, water behaves unusually in that it expands as it gets cooler, with its minimum volume at 4°C. This behaviour of water is described as anomalous (irregular). When water freezes, its volume increases by about 8%, which is a much larger increase in volume than occurs between 4°C and 0°C.
The change in volume/density of water with temperature is shown in figures 15.9 (a) and (b).

The importance of the anomalous expansion of water
When a pond is freezing over, water at 4°C, which is denser, remains at the bottom of the pond. The less dense (but lower temperature) water, between 3°C and 0°C, floats in layers above it. The water on the surface is frozen but floats because it is less dense than the water below it. The different density layers stop convection currents from spreading the heat.
Ice is a bad conductor of heat, so the layer of ice on the top of a pool acts like an insulating blanket and slows further loss of heat from the water below.
Aquatic animals and plants make use of this phenomenon by living in the liquid layer when the water freezes over in the winter.
15.4 Expansion in gases
Experiment 15.5 To show expansion in gases
Apparatus: A round bottom flask fitted with glass tube, water, trough, and source of heat from Bunsen burner.
Procedure
- Fill a trough with clean water.
- Pass a glass tube through a cork and invert the cork into a round bottom flask.
- Dip the tube in the water in the trough.

- Heat the flask by directing a Bunsen burner flame to it for a short time.
- Observe the water level in the tube.
- Allow the flask to cool while the tube is still in the water. Observe what happens.
Observation: When the flask is hot, the level of the water in the glass tube falls and air bubbles are released. On cooling, the water level rises up the glass tube.
Explanation
- When the flask is heated, the air inside expands. This forces some air out of the flask. Thus, bubbles of air are seen as the air escapes.
- As the air in the flask cools, it contracts creating more space and reducing the pressure. Then the atmospheric pressure acting on the surface of the water pushes water into the glass tube.
15.5 HEAT AND TEMPERATURE
Heat and measurement of temperature
Heat – is a form of energy which when absorbed by an object makes it hotter and when lost by an object leaves it colder.
Temperature – is the degree of hotness or coldness of a body.
Words such as warm, hot, tepid, cool, and cold tell us about the temperature of an object. These words are not very precise, so if we need to be more accurate about the temperature of an object, we use a thermometer graduated with some scales called temperature scales.
NB: Temperature is the measure of how hot or cold a body is and should not be confused with the amount of heat the body contains.
(a) Thermometers
Definition: A thermometer is an instrument used to measure temperature.
A number of different types of thermometers are available. Each type of thermometer makes use of a particular thermometric property, i.e., a property that changes with temperature. Examples of such properties are:
(i) Change in length of liquid column.
(ii) Increase in electrical resistance.
The most common type of thermometer is the liquid-in-glass thermometer. Thermometric liquids used in liquid-in-glass thermometers are:
- Mercury and alcohol.
(b) Properties of thermometric liquids
A thermometric liquid should have the following properties:
(i) Should be opaque for easy reading.
(ii) Good conductor of heat.
(iii) High and uniform expansivity.
(iv) High boiling point.
(v) Low freezing point.
(vi) Should not wet glass.
Mercury and alcohol compared
Mercury | Alcohol |
(i) Opaque and therefore can easily be read. | – Not opaque but can be coloured. |
(ii) Good conductor of heat; sensitive to small temperature changes. | – Poor conductor of heat; not sensitive to small temperature changes. |
(iii) Has uniform expansivity. | – Expansion not so regular. |
(iv) Has high boiling point (357 °C) and therefore can measure high temperatures. | – Low boiling point (78 °C) and therefore not suitable for measuring high temperatures. |
(v) Not very low freezing point (-39 °C); therefore cannot measure temperatures below its freezing point. | – Low freezing point (-115 °C). |
(vi) Does not wet glass. | – Wets glass. |
(c) Advantages of alcohol over mercury as thermometric liquid
- It has higher expansivity (about six times) that of mercury.
- It has a lower freezing point than mercury. Alcohol freezes at -115 °C while mercury freezes at -39 °C. The high freezing point of mercury makes the measurement of temperatures lower than -39 °C impossible.
Apart from the disadvantages, mercury is preferred to alcohol for the following reasons:
(i) It is a better conductor of heat than alcohol and therefore responds more readily to changes in temperature.
(ii) It is opaque and makes reading easy.
(iii) It has a high boiling point, 357 °C, whereas alcohol has a low boiling point, 78 °C, and can easily vaporize to fill the upper part with vapour.
(d) Reasons why water is not used as a thermometric liquid
Water is unsuitable for use in thermometers because of the following reasons:
(i) It freezes at 0 °C.
(ii) It has irregular expansion.
15.41 Structure of a liquid in glass thermometer
A liquid-in-glass thermometer consists of the following features:

Stem: made of uniform diameter glass capillary with a fine bore, which increases the sensitivity of the thermometer. The stem carries the temperature scale from which temperature readings are taken.
Bulb: The bulb contains the bulk of the thermometric liquid.
(f) How to use a thermometer
The bulb is inserted or placed in contact with the object/substance whose temperature is to be measured.
The liquid in the bulb will either:
(i) Acquire heat energy from the substance and expand, making its level rise; or
(ii) Lose heat energy to the substance, making its level fall.
The temperature value is then read at the liquid level which directly corresponds on the temperature scale.
15.42 Graduation or Calibration of a thermometer
In order to establish a temperature scale, we choose two fixed points. A fixed point is a definite temperature at which a change of state occurs.
The two fixed points chosen are called lower fixed point and upper fixed point.
(a) Lower fixed point – is the temperature of pure melting ice at standard atmospheric pressure (i.e., at pressure 760 mmHg).
(b) Upper fixed point – is the temperature of steam from pure water boiling under standard atmospheric pressure.
NB: The ice and the water must be pure because the presence of impurities lowers the melting point of ice and elevates the boiling point of water.
Finding the fixed points of a thermometer
(a) Lower fixed point
- Freeze some pure (distilled) water.
- Crush the ice into small pieces and fill a filter funnel with the pieces and wait for the ice to begin melting.
- When the ice begins to melt (i.e., at 0 °C), insert the bulb of the thermometer so that it is covered with ice, figure 15.11 (a). The melting ice cools the mercury to 0 °C.
- When the mercury stops shrinking (i.e., when the level becomes constant), mark the stem of the thermometer at the mercury level.
- This point is the lower fixed point, or the ice point on the Celsius scale.
(b) Upper fixed point
- Remove the thermometer and insert it through a two-holed cork.
- Fill a flask with pure water and cork it with the cork carrying the thermometer and a delivery tube such that the bulb is just above the water surface, figure 15.11 (b).
- Heat the water in the flask to boiling point.
- When the mercury stops expanding (i.e., when the level becomes constant), mark its level on the thermometer stem.
This point is the upper fixed point, or steam point (100 °C) on the Celsius scale. Then divide the difference between the two points into 100 equal points. Mark the points as a scale along the stem either in Celsius scale or Kelvin or both.

15.5 Temperature Scales
There are two common scales of temperature, namely:
(i) Celsius Scale and
(ii) Kelvin Scale
(a) Celsius scale (Centigrade)
The centigrade or Celsius scale assigns a value of 0 °C to the freezing point and 100 °C to the boiling point of pure water. It is defined by dividing the fundamental difference (the difference between the upper and lower fixed points) into 100 °C equal degrees. The temperatures on this scale are called “Degree Celsius”.
(b) Kelvin scale (Thermodynamic Scale)
In the Kelvin or absolute scale, the lower fixed point is 273.15 K and the upper fixed point is 373.15 K. The Kelvin (K) is the S.I unit of temperature.
Note:
- The degree intervals are identical to those measured on the Celsius scale.
- Absolute zero is approximately -273.15 °C or zero (0 K) degree on the Kelvin scale.
- Absolute zero is the lowest possible temperature. It is characterized by complete absence of heat energy.
- Temperatures on Kelvin do not have degree symbol (°).
- One Kelvin is the same as one Celsius, i.e., 1 K = 1 °C.
(a) Relationship between Celsius scale and Kelvin scale
The two temperature scales are related to each other as shown below.
Fixed point | Celsius scale | Kelvin scale |
| Lower fixed point | 0 | 273 |
| Upper fixed point | 100 | 373 |
The temperature value, q, on Celsius scale is related to the temperature value, T, on Kelvin scale by the formula:
q = T – 273
Or T = q + 273
Worked Examples
Convert the following temperature readings to Celsius scale:
- 1010 K
- 233 K
- 373 K
Convert the following temperature readings to Kelvin scale:
- 240 °C
- 30 °C
- 120 °C
Solution
q = ?, T = 1010 K; (b) q = ?, T = 233 K; (c) q = ?, T = 373 K
q = T – 273
= 1010 – 273 = 233 – 273 = 373 – 273
q = 737 °C, -40 °C, 100 °C
(a) q = 240 °C, T = ?
(b) q = 30 °C, T = ?
(c) q = 120 °C, T = ?
T = q + 273
= 240 + 273 = 513 K
= 30 + 273 = 303 K
= 120 + 273 = 393 K
(c) Calculating the temperature values when the lengths of the thermometric liquid for the lower and upper fixed points are given

From the diagram:
l0 = the length of mercury column at 0 °C.
l100 = the length of mercury column at 100 °C.
lq = the length of mercury column at unknown temperature, q °C.
x = (lq – l0)
y = (l100 – l0)
Temperature, q, in °C = 
= 
q = 
Worked Examples
1. A mercury thermometer is calibrated by immersing it in melting pure ice and then in boiling pure water. If the mercury columns are 6 cm and 16 cm respectively, find the temperature when the mercury column is 8 cm long.
l0 = 6 cm, l100 = 16 cm, lq = 8 cm, q = ?
q = 
= 
= 
= 20 °C
2. The length of mercury column of a thermometer at ice point and steam point are 2.0 cm and 22.0 cm respectively. The reading of the thermometer when the mercury column is 9.0 cm long is:
A. 45.0 °C B. 40.9 °C C. 35.0 °C D. 31.8 °C
l0 = 2.0 cm, l100 = 22.0 cm, lq = 9.0 cm, q = ?
q = 
= 
= 
= 35 °C
Therefore the answer is C.
Other types of thermometers
Other types of thermometers include:
- Clinical thermometer – Used by doctors in hospitals and clinics.


Constriction – Causes break to the mercury thread and stops the thread above it from moving back into the bulb. It enables the doctors to take the reading at their own time.
NB: Before use on another patient, the thermometer is shaken to let the mercury thread move into the bulb.
- Thermocouple
- Resistance thermometer
- Thermistor
- The constant-volume gas thermometer
- The maximum fixed thermometer
Self-Check 15.0
1. The distance between the fixed points on mercury-in-glass thermometer is 25 cm. What is the temperature in degrees Celsius if the mercury thread is 8 cm long?
A.  B.
B.  C.
C. 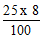 D.
D. 
2. Which one of the following fluids is the best conductor of heat?
A. Air B. Alcohol C. Water D. Mercury
3. The graph in the figure shows water being heated from –10°C to 100°C.

At what point does the substance have maximum density?
A. E
B. C
C. D
D. B
4. A bimetallic strip operates on the principle that metals
A. are heat conductors.
B. are good heat conductors.
C. have different rates of expansion.
D. have the same rates of expansion.
5. In order to make a mercury thermometer more sensitive, the
A. degree markings must be further apart.
B. diameter of capillary tube must be reduced.
C. volume of the mercury bulb must be reduced.
D. capillary tube must be open to air.
6. A tight bottle top becomes easier to unscrew when hot water flows over it because the
A. cap expands more than the glass.
B. glass in the neck of the bottle contracts.
C. hot water acts as oil between the glass and the bottle.
D. increased pressure of the air in the bottle causes the cap to expand.
7. Which of the following changes occur when a metal block is heated?
| Volume | Mass | Density | |
| A | increases | remains the same | decreases |
| B | increases | increases | increases |
| C | remains the same | remains the same | decreases |
| D | increases | remains the same | increases |
8. The distance between the lower and the upper fixed points on the Celsius scale in unmarked mercury-in-glass thermometer is 25 cm. If the mercury level is 5 cm below the upper fixed point, then the temperature is:
A. 5°C B. 20°C C. 80°C D. 95°C
9. The unusual expansion of water when it is cooled between 4°C and 0°C is due to:
A. Water molecules coming closer together to form a compact structure.
B. Formation of a new arrangement of molecules which requires a large volume.
C. The increased repulsive forces between the water molecules.
D. Differences in the sizes of water and ice molecules.
 10. Which one of the following graphs shows how the density of water varies with temperature between 0°C and 100°C?
10. Which one of the following graphs shows how the density of water varies with temperature between 0°C and 100°C?
SECTION B
(a) Define the following terms:
(i) Expansion
(ii) Contraction of a material
(b) Describe an experiment to demonstrate expansion and contraction in solids.
(c) State any one application of expansion in solids.
(a) Define linear expansivity of a material.
(b) Calculate the linear expansion of a concrete bridge of span 100 m when the temperature rises by 20°C. The linear expansivity of concrete is 1.2 × 10-5/°C.
(a) Describe an experiment to show expansion in liquids.
(b) Explain the anomalous behaviour of water and give its importance to aquatic animals.
(a) State:
(i) Any two thermometric liquids you know.
(ii) The properties of a thermometric liquid.
(iii) Advantages of mercury over alcohol as used in a thermometer.
- Define the following terms.
(i) Lower fixed point.
(ii) Upper fixed points.
(c) Describe how the fixed points of a thermometer are determined in the laboratory.
5. The interval between the ice and steam points on a thermometer is 192 mm. Find the temperature when the length of the mercury thread is 67.2 mm from the ice point.
6. The distance between the lower and upper fixed points on the Celsius scale in unmarked mercury-in-glass thermometer is 25 cm. If the mercury level is 5 cm below the upper fixed, calculate the temperature value.
7. Convert the following temperature readings to Celsius scale:
(a) 750 K (b) 400 K (c) 973 K
8. Convert the following temperature readings to Kelvin scale:
(a) 340 °C (b) 130 °C (c) 20 °C
9. (a) Name any two physical properties which change with temperature.
(b) Explain why gaps are left between rails in a railway line.
(c) Why do gases expand much more than solids for the same temperature change?
(d) Name one application of a bimetallic strip.
(e) Mention any three reasons for not using water as a thermometric liquid.
10. Figure 15.13 shows strips of copper and iron bonded together.

(a) Redraw the diagram to show what happens when the strip is heated.
(b) Why does the change you have shown in (a) take place?
ANSWERS TO SELF-CHECK QUESTIONS
| CHAPTER | SELF-CHECK NUMBER | NUMERICAL ANSWERS |
| 2 | 2.0 | 1. (b) 24.8 mm or 2.48 cm or 0.0248 m (c) (i) 4.27 cm or 0.0427 m (ii) 10.63 cm or 0.1063 m |
| 2. (b) (i) 6.68 mm (ii) 7.47 mm (iii) 9.74 mm | ||
| 2.1 | 1. (a) (i) 10 km (ii) 2,000 cm (b) (i) 25,000 g (ii) 2 kg (c) (i) 43,200 s (ii) 900 s (d) (i) 0.02 m³ (ii) 50,000,000 cm³ or 5 × 10⁷ cm³ | |
| 2.2 | 1. (a) 3 (b) 1 (c) 4 (d) 4 (e) 1 2. 34.1 mm 3. (a) 15 m² (b) 80 m³ | |
| 2.3 | (a) (i) 2.22 × 10⁻⁴ (ii) 2.5 × 10⁻³ (b) (i) 5.62 × 10³ (ii) 7.5 × 10⁴ (c) (i) 1.2 × 10¹¹ (ii) 2 × 10¹ (d) (i) 5 × 10⁰ (ii) 1.1 × 10³ (e) (i) 2.5 × 10⁴ m (ii) 2.5 kg (iii) 11 × 10⁻² | |
| 2.4 | 1. A 2. B 3. D 4. C 5. D 6. B 7. B 8. D 9. D 10. B 11. B 12. B 13. D 14. A 15. B | |
| 3 | 3.0 | 1. D 2. D 3. C 4. B 5. C 6. C 7. D 8. B 9. A 10. B 11. A 12. A 13. B 14. B 15. B 16. D 17. B 18. D 19. A 20. C |
| … |






