SECOND TERM E-LEARNING NOTE
SUBJECT:BASIC SCIENCE CLASS:JS 3
SCHEMEOFWORK
WEEK TOPIC
- Electrical Energy
- Skill Acquisition
- Teenage Pregnancy and Abortion
- Elements, Compound and Atomic Structure
- Radioactivity
- Chemical Formula
- Writing and Balancing Chemical Equations.
- Simple Machines
- Digestive System
- Circulatory System
REFRENCES
- Basis Science Made Easy for JSS Three by F.I Kehinde et al
- Basis Science Made Easy for JSS Two by F.I Kehinde et al
- STAN Integrated Science for JSS Three
- Precious Seeds Basic Science for JSSThree by J.O Otugboyega et al.
WEEK ONE
TOPIC: ELECTRICAL ENERGY
CONTENT
- Introduction.
- Concept of Electron Flow.
- Concept of Current, Resistance and Potential Difference.
- Series and Parallel Arrangement in Electric Circuit.
- House Circuit: Fuse and Circuit Breaker.
- Electric Meter Reading and Billing.
INTRODUCTION
Electricity is the flow of electric current. Electric Current is the continuous flow of charges (electrons). It is also the rate of flow of charges. Electricity has several applications in the modern day society. Electrical energy is needed by industries, hospitals, offices, homes, schools, research institutes and so on. Power generation in Nigeria is by electricity Distribution Company of Nigeria (EDCN), which is saddled with responsibility of generating power, distribution of power and billing of power consumption.
Concept of Electron Flow
When current is flowing in a wire, electrons are moving rapidly. Electrons have negative charges. Negative charge is usually shown with a minus sign. Electrons flow round a path called circuit.
Concept of Current, Resistance and Potential Difference
Current: current is the continuous flow of electrons. It is also defined as the rate of flow of charges. It is represented as I. It measured in ampere (A). It is measured using an instrument called ammeter. Depend on the source of generation, there are two types of current: Direct current (DC) and alternating current (AC). Direct current is gotten from small generators, batteries and inverters while the alternating current comes from the transmission lines and transformers.
 Resistance: this is the opposition to the flow if electric current in a circuit.Is represented as R. It is measured in ohms with an instrument called ohmmeter. An electrical component designed to introduce a known value of resistance into a circuit is called a resistor.
Resistance: this is the opposition to the flow if electric current in a circuit.Is represented as R. It is measured in ohms with an instrument called ohmmeter. An electrical component designed to introduce a known value of resistance into a circuit is called a resistor.
 Potential difference (P.D): This is the energy required to move charges from the positive terminal to the negative terminal. P.d is represented as V and measured in volt using an instrument called Voltmeter.
Potential difference (P.D): This is the energy required to move charges from the positive terminal to the negative terminal. P.d is represented as V and measured in volt using an instrument called Voltmeter.
Series and Parallel Arrangement in Electric Circuit.
Series Arrangement of Resistors.

 Series connection is the connection of resistors from one end to another.
Series connection is the connection of resistors from one end to another.
RT = R1 + R2 + R3
RT means total or effective resistance.
Parallel Arrangement of Resistors.
This is the arrangement in which resistors are arranged side by side such their corresponding ends join together at two common junctions.
1/RT = 1/R1 + 1/R2 + 1/R3
EVALUATION
- Differentiate between parallel and series arrangement.
- Define the following (a) current (b) potential difference (c) Resistance.
- What is electricity?
HOUSE CIRCUIT: FUSE AND CIRCUIT BREAKER.
220V of electricity enters our homes from the transformer and it us sent into the meter.
In order to prevent power surges or electrical problems, fuses and circuit breakers are used. A fuse is a safety device in an electric circuit. It consists of a thin resistance wire that will permit only a certain amount of electric current to pass through it, otherwise it will melt. Fuses are placed in the main electrical supply to protect wiring in the house. A circuit breaker has a switch that automatically goes off when there is an electrical problem it can be turned on later.
Billing of Electric Meter Reading
At home, electrical power is measured and billed. Power is the rate at which work is done.
Electrical power = current × voltage
Electrical power is measured in watt using an instrument known as wattmeter.
One watt is the power consumed in an electric circuit when one joule of work is done in one second. Larger units of power are the kilowatt (kW) and megawatts (MW).
1KW = 1000 watts = 103 W
1MW = 1000000 watts = 106 W= 103 KW.
In order to bill an electric Meter, electric energy consumption is measured and sold by the Electricity Distribution Company of Nigeria (EDCN) in units of kilowatt-hour.
Example: A radio set is used for 12 hours per day for 65days. Find the quality of energy consumed and the cost, if the radio set is rated 35W. Assume that the cost of 1 kWh is 50 kobo.
Solution
Quantity of energy consumed = power × time
Energy consumed= 35W × 12hours × 65days
Energy consumed= 27300Wh
 Dividing by 1000 to make it KWh = 27300Wh
Dividing by 1000 to make it KWh = 27300Wh
1000
Therefore, the quantity of energy consumed= 27.3kWh
Cost= quantity of energy consumed × the cost of 1 unit of energy
Cost of 1 unit of energy= 50kobo
Cost of energy consumed= 27.3kWh × 50kobo= 1365kobo= 13.65
Electric meter reading
Electric meter is a device installed in homes, industries, schools and offices to read the quantity of electrical energy used by local consumers of electricity. To read the meter, the previous unit is subtracted from the current unit, which will give the actual meter reading of the meter.
For example, if the previous unit or reading on the meter is 48,626.8 units and the current reading is 59,014.1 units, the actual meter reading for the month will be given as follows:
Current reading – Previous reading = Actual reading
50,014 – 48,626.8 = 1,387.3
Therefore, the meter reading for the month is 1387.3
EVALUATION
- State the difference between fuse and circuit breaker.
- What device is used to measure electrical power?
GENERAL EVALUATION/REVISIONAL QUESTIONS
- What is electrical power?
- State the use of: (a) ohmmeter (b) ammeter (c) wattmeter
- What is work?
- State the features that adapt (a) birds (b) fishes to their habitat
- What is genetic counselling?
READING ASSIGNMENT
Precious Seeds Basic Science for JSS three by J.O otugboyega et al. Chapter 16. Pg 99-101
WEEKEND ASSIGNMENT
- The instrument used for measuring electric current is A. Ammeter B. Wattmeter C. Voltmeter D. Ohmmeter
- The kind of current generated by EDCN is _____ current A. Parallel B. Series C. Direct D. alternating
- ___ is the opposition to electric current A. Potential difference B. Electrical power C. Resistance D. Fuse.
- A device which is used to prevent power surges from damaging appliances is called A. Meter B. Fuse C. Coil D. Wattmeter
- The type of connection commonly used at home is ____ A. Parallel B. Series C. Direct D. alternating
THEORY
- What is electricity?
- Explain electron flow.
WEEKTWO
TOPIC: SKILL ACQUISITIONS
CONTENT
- Meaning Skill Acquisition
- Reasons for Skill Acquisition
- Types of Skill
- Importance of Skill Acquisition
Meaning of Skill Acquisition
A skill is a special ability acquired through training in order to do something in a better way. Skill is also defined as the ability to bring about some end result with maximum certainty an minimum outlay of time and energy. There is a different between talent and skill. Skill is usually acquired through training from either a formal or an informal setting. Talent is an inborn ability to do certain things. It is possible to be talented but not skilled. The training acquired will help the talented individual to improve on his/her ability. An acquired skill is perfected through constant practice and training. Psychologists believe that when you practice a skill continuous for seven years or 10,000 hours it becomes part of you. Someone who has undergone training to acquire a skill is called a professional or an expert while someone who knows little or nothing about a skill is called a novice. The process of moving from being a novice or unskilled person to a skilled person or a professional is called skill acquisition. Skill acquisition is the process of learning and mastering a particular skill.
Reasons for Skill Acquisition
- To earn a living: Skill could be acquired in order provide a means of livelihood for the individual. The individual uses the money earned from using his skills to meet basic needs.
- For employment: Acquiring a skill provides an individual with the opportunity of being self-employed. Skill acquisition reduces the stress of seeking for a job all over the place. The more engaged a person is in a profession, the more skillful he/she becomes. Such a person can even earn more money by training others. The person becomes a decision maker and a manager of emergencies
- For independence: skill acquisition enables one to be independent, not relying on friends and family for survival
Other reasons for skill acquisition include:
- Risk taking
- Decision making
- Managing emergency situation
- Survival strategy
- Learning to live together
- Improvement of quality of life.
- Appreciating human capacity
EVALUATION
- What is skill?
- What are the reasons for skill acquisition?
Types of Skills
There are different skills that can be acquired and they include:
- Automobile repairs (mechanical works)
- Painting
- Hair dressing/barber’s work
- Photography
- Publishing
- Catering
- Panel beating
- Book binding
- Farming
- Computer literacy
- Fine art
- Welding
- Brick laying
- Plumbing
- ICT
- Metal work
- Electrical installation
- Video coverage
- Fish farming
- Trading
- Shoe making
- Fashion designing
- Upholstery/carpentry
- Hat/bead making
- Desktop publishing
Importance of Skill Acquisition
- Improvement in skills and knowledge.
- Improvement in communication.
- Improvement on the quality of life.
- Survival strategy.
- Means of livelihood.
- Self-gratification and independence.
- Meaningful contribution to the society.
Evaluation
- State five importance of acquiring skills.
- State ten skills that can be acquired.
GENERAL EVALUATION
- State ten types of skills.
- What are the reasons for acquiring skills?
- State four cause of flooding.
- What is light energy?
- State the laws of refraction.
READING ASSIGNMENT
Basis Science Made Easy for JSS Three by F.I Kehinde et al. Chapter 14. Page 61-64
WEEKEND ASSIGNMENT
- A special ability acquired through training is called A. Method. B. Process. C. Entertainment. D. Skill.
- One of the following is not an acquired skill A. Photography B. Sleeping C. Catering D. Weaving
- Someone who has acquired a skill is called a A. Professor B. Cohesion C. ProfessionD. Professional
- Which of the following is not a reason for acquiring skills? A. Survival strategy B. Dependency on friends C. Self-employment D. Training others
- A person who has little or no knowledge about a particular skill iscalled A. Professional B. Expert C. Guru D. Novice
THEORY
- Define skill acquisition.
- Mention five types of skills.
- State four reasons for acquiring skills.
- State four importance of skill acquisition.
WEEKTHREE
TOPIC: TEENAGEPREGNANCYANDABORTION
CONTENT
- Meaning of Teenage Pregnancy
- Consequences of Teenage Pregnancy and Delivery
- Myths and Facts About Pregnancy
Teenage pregnancy refers to pregnancy in a girl between the ages of 10-19. Teenage pregnancy is usually unintended and unwanted. The fact that you are at pubertal age and can get pregnant physically does not mean that you are really mature to be pregnant socially, emotionally, educationally even financially.
Teenage pregnancy can be dangerous for both the mother and the unborn child. Complication at child birth, and unsafe abortion, usually result from teenage pregnancy.
CONSEQUENCES OF TEENAGE PREGNANCY AND DELIVERY
The following are some of the consequences of a girl who got pregnant at the unripe age of teens.
1. Health Risks: Pregnancy induced hypertension, which can cause heart failure and death of both mother and the child. This type of pregnancy can lead to a condition whereby the girl’s pelvic opening is too small to allow the infants head to pass during delivery. This may cause tear between the vagina and the urinary tract (vesico vagina fistula) or rectum(recto vagina fistula). Caesarian section (C.S) may result because the hips are not widened enough for the baby to come through the pelvis.
2. Unsafe Abortion: Since teenage pregnancy is unwanted, they are likely to seek abortion from untrained health care providers or can go on self-medication for self-induced abortion.
3. Socio-economic:
- Termination of education.
- Stigmatization
- Life span and career goals are disrupted
- Early marriage due to pregnancy
- Isolation from peers and low self esteem
- Low income and economic dependence
- Force marriage
- Poverty
- Emotional feeling of guilt and fear
- Loose of childhood and adopting to adulthood.
MYTHS AND FACTS ABOUT PREGNANCY
- Myth: Girls should not use contraceptives.
Fact: Girls should abstain from sexual intercourse so as to prevent themselves using contraceptives but if they have sexual intercourse and use contraceptive, it is safe and can prevent teenage pregnancy.Thus contraceptives are not for bad girls.
- Myth: The contraceptives causes the following:
- It destroys the womb.
- It makes one infertile for life.
- It makes one thin.
- It reduces one’s breast size.
Fact: Only a few contraceptive may cause delay in fertility. Contraceptives do not damage the womb but drug overdose or abuse and unsafe abortion may damage the womb.
- Myth: The condom reduces sensitivity during sexual intercourse.
Fact: There are very thin latex condoms and textured condoms that favour sensitivity in sexual relation.
- Myth: A woman can get pregnant as a result of kiss.
Fact: Spermatozoa must penetrate the vagina for fertilization to take place somewhere in fallopian tube.
- Myth: It is better for a woman to have children while still young as she can see them grow.
Fact: The ideal age for pregnancy is between 25 and 35.
- Myth: For a woman to obtain the love of a man, she must have child for him.
Fact: Pregnancy does not guarantee a good relationship. A partner can leave even after there is a child in the relationship.
EVALUATION
- Mention five reasons why you will not engage in sexual intercourse now as a JSS 3 student.
- Mention five ways teenage pregnancy can be prevented
CONTENT
- Meaning of Abortion
- Types of Abortion
- Reasons for Abortion
- Unsafe abortion and it Consequences
- Ways to Prevent Abortion
- Where to get help
Meaning of abortion
Abortion is the deliberate termination of a human pregnancy, most often performed during the first 28 weeks. It is the ending of pregnancy by removing a foetus or embryo before it can survive outside the uterus. An abortion that occurs spontaneously is also known as a miscarriage. An abortion may be caused purposely and is then called an induced abortion.
TYPES OF ABORTION
There are two main types of abortion and they are;
(i) SpontaneousAbortion(ii)Induced Abortion
Spontaneous Abortionis an unintentional abortion caused as a result of faulty development of embryo, abnormality of placenta, hormonal disturbances, acute infection, severe trauma or shock. It is commonly called miscarriage.
Induced Abortionis the intentional removal of foetus. Induced abortion can either be surgical or medical.
(a) Medical Abortion: This is the abortion done or induced with the use if one or more pills
(b) Surgical Abortion: This form involves minor surgical operation to induce abortion.
REASONS FOR ABORTION
The following are some of the reasons why people engage in abortion:
- Teenage pregnancy or unwanted pregnancy.
- Pregnancy as a result of rape.
- Poverty and lack of financial capacity to care for the unborn child.
- Medical complication at the onset of the pregnancy that may endanger the life if the mother or the foetus.
- Lack of readiness.
UNSAFE ABORTION AND ITS CONSEQUENCES
An unsafe abortion is the abortion done without proper medical supervision. Unsafe abortion is usually by using over-the-counter drugs or carried out by quack medical practitioners.
Teenagers are lured to unsafe abortion for the following reasons:
- The fears of letting someone know about their pregnancy.
- To avoid social stigma.
- To protect their education and future.
As convincing as they sound, these reasons don’t weigh up to the consequences that comes with unsafe abortion. The following are the consequences of unsafe abortion:
- Unsafe abortion may lead to the damage of internal organs like the womb.
- It is painful and horrible.
- Teenager can get infected from the unsafe process of abortion which may cause more severe disease like uterine fibroid.
- It leads to the death of the teenagers.
- It endangers the teenager as the quack doctor may further molest her.
- There is a psychological breakdown if the teenager cannot endure going through the abortion.
EVALUATION
- What is abortion?
- State four consequences of unsafe abortion.
WAYS TO PREVENT ABORTION
The following can help prevent abortion or reasons to abortion:
- Abstinence: Teenagers should avoid having any until they are physically and psychologically matured to take responsibility.
- Avoid unsafe and unprotected by using condoms.
- Women should go for medical checkup to prevent medical complication during pregnancy.
- Keep responsible friends and partner.
- Create a mindset against abortion.
- Meet your school counselor for advice on abstinence.
- Avoid sex promoting/pornographic content.
- Always remember that the negative consequences of abortion outweigh the benefits.
WHERE TO GET HELP
Teenagers need help on what to do when faced with abortion related challenges. Here are some of the things you can do when faced with a challenge:
- Talk to your parents about the about the situation
- Meet a counselor for an advice on what to do.
- There are also teenagers-support NGOs that can help.
- Join a school social club against teenage pregnancy and abortion. E.g. anti AIDS club.
- Join a true religious or social body that can help with teenage pregnancy and abortion related issues.
EVALUATION/REVISIONALQUESTIONS
- Mentionsevenimplicationsofteenagepregnancy
- Whatarehormones?
- Whatisabortion?
- Mentionfourreasonsforengaginginabortion.
- Explainwhylighteningisseenbeforethunder.
- Whatisamachine?
READING ASSIGNMENT
Basic Science for Junior Secondary School by J.O. Otugboyega et al. Page 127-130
WEEKEND ASSIGNMENT
- The type of abortion that involves taking over-the-counter drugs is (a) Traditional (b) Medical (c) Surgical (d) None of the above
- A developing child in the womb is called a (a) Baby (b) Foetus (c) Zygote (d) None of the above
- Which of the following is not a way of preventing abortion (a) living reckless life (b) Abstinence (c) safe sex (d) None of the above
- Where does the foetus develop (a) fallopian tube (b) uterus (c) vagina (d) None of the above
- A person who constantly performs illegal abortion is called (a) Oncologist (b) Therapist (c) Abortionist (d) None of the above
THEORY
- Explain with reason why abortion should or should not be legalized in Nigeria.
- What advice will you give to a pregnant teenager based on your knowledge of the topic?
WEEK FOUR
TOPIC: ELEMENTS, COMPOUND AND ATOMIC STRUCTURE
CONTENT
- Element
- Compound
- Formula of Binary Compound
- Fundamental Particles of Atom
Element
An element is a substance which cannot be further divided into simpler substances. Over 100 chemical elements are known. They fall into two main categories, namely: Metals and Non-metals.
Metallic elements have their ending with letter M while non-metal element may not have named ending with letter M but with other alphabets except very few.
Compound
Chemical compounds are formed when two or more elements combine together. On this basis, two major categories of chemical compounds are known: (a) Binary Compound (b) Non-Binary Compounds.
BINARY COMPOUNDS | NON-BINARY COMPOUNDS |
Their names end with-ide | Their names often end with-ate |
They are made of only two elements | They are made of 3 or more elements |
EXAMPLES OF ELEMENTS AND SYMBOLS
Hydrogen H Sodium, Na
Helium, He Magnesium, Mg
Lithium, Li Aluminium, Al
Berilium, Be Silicon, Si
Boron, B Phosphorous, P
Carbon, C Sulphur, S
Nitrogen, N Chlorine, Cl
Oxygen, O Argon, Ar
Flourine, F Potassium, K
Neon, Ne Calcium, Ca
EXAMPLES OF COMPOUNDS AND FORMULA:
Water H20
Sand Si02
Chalk CaC03
Lime CaO
The elements in a chemical compounds cannot easily be separated for they are chemical combined. The ratios of combination of atoms of these elements in compounds are fixed unlike in mere mixture.
EVALUATION
- Define (a) element (b) Compound.
- What are binary compounds?
- State five examples each of metals and non-metals.
FORMULAE OF BINARY COMPOUNDS
Binary Compounds are compounds of elements only and names ending with-ide. E.g. Magnesium chloride.Magnesium Chloride = Magnesium + Chlorine.
Elements | Atomic Electronic Number | Group Configuration | Valency | Oxidation number |
H | 1 | 1 | 1 | +1 |
He | 2 | 2 | 0 | 0 |
Li | 3 | 2,1 | 1 | +1 |
Be | 4 | 2,2 | 2 | +2 |
B | 5 | 2,3 | 3 | +3 |
C | 6 | 2,4 | 4 | +4 |
N | 7 | 2,5 | 3 | -3 |
O | 8 | 2,6 | 2 | -2 |
F | 9 | 2,7 | 1 | -1 |
N | 10 | 2,8 | 0 | 0 |
Combinations of elements are made on the basis of their combining Capacities called valencies. Besides, every atom of element contain electrons by which distribution in a given order termed ELECTRONIC
CONFIGURATION, the group and hence valency of each element in combination is decided. For example, Magnesium nitride.
Mg = 12 = 2, 8, 2 = Group = 2, valency = +2
N = 7= 2 , 5 = Group = 5, valency = -3

 Mg N = Mg3 N2 = Magnesium nitride.
Mg N = Mg3 N2 = Magnesium nitride.
2 3
EVALUATION
- What is electronic configuration?
- Define the term Valency.
- What is the valency of an element found in group 6?
- Work out the formulae of Aluminum oxide and carbon dioxide.
Fundamental particles of Atom
The atom is made up of a number of different particles.
There are three fundamental units namely;
- The electron (discovered 1897)
- The proton (discovered 1920)
- The neutron (discovered 1932)
THE ELECTRON
The electron carries a negative electric charge and indeed a very small function of the mass of even the smallest atom, hydrogen.
They are at a comparatively great distance from the centre of the atom and travel rapidly and continuously in orbits around its centre known as the nucleus.
The electrons were discovered first in 1897 by Sir J.J. Thomson.
THE PROTON
The proton is a positively charged particle found in the nucleus of an atom although the positive charge on the proton has same magnitude charge on the electron mass of the proton which is about 1800 times larger than that of the electros.
The number of protons in the nucleus of an atom is called atomic number. All atoms of the same element have the same atomic number but those of different elements have a different number of protons in their nuclei.
The sum of the protons and neutrons in the nucleus of an atom is called the mass number with the symbol. The proton was discovered by Sir, Ernest Rutherford in 1920.
THE NEUTRON
The neutron was discovered in 1932 by a British Scientist, James Chadwick. It has almost the same charge.
Whenever the neutron number in the nucleus of an atom of the same element varies, its mass number also varies and hence we have ‘Isotopes’.
Isotopes are atoms of elements of the same atomic number but different mass number because of the difference in their neutrons number. The three common isotopes of hydrogen are:
- Proton – 1 proton
- Deuterium – 1 Proton + 1 neutron
- Tritium – 1 Proton + 2 neutrons
The particles proton and neutrons which make up the nucleus are – called Nucleons

EVALUATION/REVISIONAL QUESTIONS
- What are isotopes? Name the three isotopes of hydrogen.
- How many neurons are present in the following atoms?
- 23Na, (b) 31P, (c) 39K (d) 35Cl.
- Name the three fundamental particles of an atom.
- What is light energy?
- State the difference between echo and reverberation.
READING ASSIGNMENT
Basis Science Made Easy for JSS Three by F.I Kehinde et al. Chapter 12. Page A43-44.
WEEKEND ASSIGNMENT
- A substance which cannot be further divided into simpler substances is…………(a) Elements (b) Compound (c) Radical (d) Mixture
- One of these is NOT a binary compound ……..(a) Sodium chloride (b) Magnesium nitride
(c) Aluminium oxide (d) Calcium Phosphate
- The positively charged sub atomic particle is (a) element (b) electron (c) neutron (d) proton
- ______ discovered the electron (a) James Chadwick (b) Sir, Ernest Rutherford (c) Sir J.J. Thomson (d) Issac Newton
- The sub atomic particle involved in chemical reaction is (a) electron (b) neutron (c) shell
(d) proton
THEORY
- Explain the term ‘atom’.
- Differentiate between a binary compound and non-binary compound.
WEEKFIVE
TOPIC: RADIOACTIVITY
CONTENT
- Meaning of Radioactivity
- Discovery of Radioactivity
- Types of Radioactivity
- Radioactive Elements
- Types of Radiation and Their Properties
- Nuclear Transmutation
- Uses of Radioactivity
Meaning of Radioactivity
Radioactivity is the spontaneous disintegration or breaking down of the nucleus of an atom which makes it give out or emit radiation. Any element that disintegrates spontaneously and release rays us called a radioactive element
Discovery of Radioactivity
Radioactivity was discovered by a French physicist called Henri Becquerel in 1896. He discovered that atoms of some elements gave out some kinds of ray which penetrated a thick black paper. He used uranium for his study. His discovery was confirmed by the couple, Merie and Pierre Curie.
Types of Radioactivity
There are two types of radioactivity. i. Natural radioactivity ii. Artificial radioactivity.
Natural radioactivity occurs on its own without man’s influence.
Artificial radioactivity is caused by man’s activity.
Radioactive Elements
Radioactive elements are elements that give out rays from the nuclei of their atoms. Natural radioactive elements include uranium, radium, plutonium, radon, thorium etc. Artificial radioactive elements are produced by bombarding nonradioactive elements (like sulphur-35, iodine-137, cobalt-60 etc.) with radioactive particles to form radioactive isotopes.
EVALUATION
- What is radioactivity?
- What are the types of radioactivity?
- What are radioactive elements?
Types of Radiation and Their Properties
There are three types of rays, they are; i. Alpha particles ii. Beta particles iii. Gamma rays
Alpha particles (α-rays)
Alpha particles or rays are helium nucleus consisting of two protons and two neutrons (I.e. atomic number and atomic mass are 2 and 4 respectively).
- Alpha particles are positively charged.
- They have the heaviest radiation.
- They have the lowest penetrating power and can be stopped by a thin sheet of paper.
- They have the highest ionization power.
Beta particles (β-rays)
Beta particles or rays consist of a stream of fast moving electrons.
- They are heavier than gamma rays.
- They are negatively charged.
- They have higher penetrating power than alpha rays. They can be stopped by aluminum foil.
Gamma rays (ϒ-rays)
Gamma rays are electromagnetic radiation.
- They do not carry any charge.
- They have negligible mass.
- They have the highest penetrating power and can only be stopped by lead block.
- They have the lowest ionization power.
Nuclear Transmutation
The changes in the nucleus of the atom of an element caused by radioactive emission are called nuclear transmutation. For instance, Radium-226 emits -rays and turns to radon-222.
22688Ra—>42He + 22286Rn
If the changes in the nucleus are caused by man, it is called artificial nuclear transmutation.
EVALUATION
- Differentiate between alpha, beta and gamma radiations.
- What is nuclear transmutation?
Uses of Radioactivity
Radioactivity has application in the following fields;
- The medical field.
- Agricultural and scientific research.
- The industrial field.
In medical field
Radioactivity is used in;
- Sterilization of syringes and medical equipment.
- Treatment of cancer.
- Sterilization of bottled drinks and canned food.
In agricultural and scientific research
Radioactivity is used as;
- Radioactive tracers.
- Radioactive or carbon dating.
In industrial field
Radioactivity is used to;
- Study defects in metals and welded joints.
- Check metal weaknesses.
- Trace underground pipe leakages.
Effect or dangers of using radiation
- Long exposure to radiation can destroy body cells.
- Exposure to radiation can lead to gene mutation.
- It can cause deformities to foetus in the womb.
- Radiation from nuclear weapons can kill.
- Radioactive wastes pollute and destroy the environment.
EVALUATION
- State the uses of radioactivity in (a) medical field (b) industries (c) agricultural and scientific research.
- List the hazards of radioactivity.
EVALUATION/REVISIONALQUESTIONS
- Whatisradioactivity?
- Whatisreproduction?
- Mention three benefitsofacquiringskills.
- Whatisozone layer depletion?
- Whatisreflection?
READING ASSIGNMENT
Basic Science for Junior Secondary School by J.O. Otugboyega et al. Page102-103
WEEKEND ASSIGNMENT
- ____ is referred to as the father of radioactivity. (a) Henri ford (b) Pierre Currie (c) Marie Curie (d) Albert Einstein
- Positively charged radiation that can be stopped by a thing sheet of paper is (a) X-ray (b) Beta particles (c) Alpha particles (d) Gamma rays
- Blood moves out of the heart to the lungs through ____ (a) Aorta (b) Pulmonary artery (c) Vena Cava (d) hepatic artery
- Which of the flowing is an electronegative element? (a) Na (b) F (c) Hg (d) Au
- Which of the following is not a result of excess exposure to radiation? (a) HPV (b) cancer (c) Mutation in foetus (d) Radioactive poisoning
THEORY
- State five benefits of radioactivity.
- Should Nigeria embark on nuclear power as alternative source of electricity? Give reasons for your answer.
WEEKSIX
TOPIC: CHEMICALSYMBOLSANDFORMULAE
CONTENT
- Symbolsof Elements.
- ElectronicConfiguration.
- Groupand Valencyof Compounds.
- Formulasof Compounds.
- IUPAC Nomenclature.
- Fundamental Particles of Atom.
SYMBOLSOFELEMENT
AnelementisasubstancewhichconsistsofonlyonekindofmatterOver100elementsareknown,theseelementsareusuallyrepresentedbysymbolsinsteadofwritingtheirnamesinfull.
TheabbreviationforeachelementiscalledSymbolforthatelement.
Thebriefnameofthatelementitrepresentsalsooneatomoftheelementitstandsfor.(Element1–20)
Thereare4groupsofsymbols:
Symbolsformedfromthefirst letterofelement
Element Symbols
Hydrogen H
Carbon C
Nitrogen N
Oxygen O
Sulphur S
Phosphorus P
Iodine I
Fluorine F
(b) Symbolsformedfromthefirsttwoletteroftheelement.
Elements Symbols
Helium He
Lithium Li
Beryllium Be
Neon Ne
Aluminum Al
Calcium Ca
Barium Ba
Bromine Br
(c) Symbolsformedfromthe first andanyletterinthenameoftheelements
Elements Symbols
Magnesium Mg
Chlorine Cl
Manganese Mn
Zinc Zn
Cadmium Cd
Platinum Pt
(d) Symbolsderivedfromlatinnameoftheelements
Element Latin Name Symbols
Sodium Natrium Na
Iron Ferrum Fe
Copper Cuprum Cu
Gold Aurum Au
Potassium Kalium K
Silver Argentun Ag
Lead Plumbum Pb
Mercury Hydragyrum Hg
EVALUATION
- Definethetermsymbolofanelement.
- Mentionthefourgroupsofsymbolthatwehave.
ELECTRONICCONFIGURATION
ElectronicConfigurationistherepresentationofhowelectronsarearranged ontheshellsofanelement.
Thepatternofhowelectronsarearrangedisofthefashion:
K 1stshell–2electrons
L 2ndshell–maximumof8electrons
M 3rdshell–maximumof8electrons
N 4thshell–maximumof8electrons
Thispatternworksforatleastthe first 20elements.
Theelectronicconfigurationhelpsustocorrectlyplaceelementsintotheirgroupsandperiodwithinperiodictable.Thisinformationalsohelpsinthedeterminationofcombiningpowersofelements.
EVALUATION
- Whatiselectronicconfiguration?
- WritetheelectronicconfigurationofNa=11,P=15.
GROUP AND VALENCY
Thegroupreferstotheverticalarrangementincolumnsofelementsintheperiodictable.Valencyisthecombiningpowerofanelement.Itmustbecorrectlydeterminedandthecorrect chemical formulaeofcompoundweretobedetermined.
Group Valency
I +1
II +2
III +3
IV –4
V –3
VI –2
VII -1
O 0
EVALUATION
1. Definethetermvalency.
2. Whatisthevalencyof(a)GroupIIIelement(b)GroupIVelement.
FORMULAEOFBINARYCOMPOUND
Formulaeofbinarycompound
Acompoundisasubstancemadeof2ormoreelementschemicallycombinedtogether.Compoundof2elementsonlyareknownasbinarycompoundsandhavethefollowingcharacteristics.
- Namesendwith–IDE.
- Composedofonly2elements.
E.g.Carbon dioxide, Magnesium chloride, AluminumOxide,AluminumChloride.
C=6______2,4 Al=13______2,8,3
Mg=12______2,8,2O=8______2,6
Cl=17 _______2,8,7



 Mg Cl A1 0
Mg Cl A1 0
+2 1 +3 2
MgC12 Al203

 Al C
Al C
+3 -4
Al4C3
NON-BINARYCOMPOUNDS
Thesearecompoundsof2ormoreelementswhosenamesendwithATEexamplesare:Magnesiumtrioxocarbonate(IV)
Berylliumtrioxonitrate(V)
Calciumtetraoxosulphate(VI)
PotassiumheptaxodichromateVI
SodiumhexachloroplatimateII
Non-binarycompoundshavethesecharacteristics:
EVALUATION
- Whatarenon-binarycompound?
- Givetwooftheircharacteristics.
- Stateanyfiveexampleofnon-binarycompound.
DETERMINATIONOFVELENCIESOFRADICALS
Radicalsaregroupofatomsbearinga charge.Theyarethepolyatomicionsi.e.ionswhichcontainmorethanonetypeofatom.Exampleinclude;
1. Trioxonitrate (V) ion N03–
2. Trioxocarbonate(IV) ion C032-
3. Tetraoxosulphate(VI) ion S042–
4. Hydroxideion OH–
5. Cyanideion CH–
6. Tetraoxophosphate (V) ion PO43-
7. Hydrogentrioxocarbonate(IV)ion HC03–
Thechargeoneachpolyatomicionisdeterminedthus:
S+4(O)
But O=-2andS=6
S+4(-2)
6–8=-2
SO42-
CO32-
C+3(O)
But C=4,O=-2
4+3(-2)
4–6=-2
C032-
FORMULAEOFNON-BINARYCOMPUNDS
SodiumhexachloroplatinateII
Na=11________2,8,1
Valencyofsodium=1
HexachlroroplatinateII=PtCl6
Pt+6Cl
2+6(-1)
2–6
=-4
ValencyofPtCl6=-4
 Na PtCl6
Na PtCl6

1 4
Na4PtCl6
EVALUATION
- Name the following (i) CaC03(ii)Na3PO4.
- Workoutthechargeonthefollowingpolyatomicions
(a)SO3n(b)NO3n.
IUPACNOMECLATURE
InternationalUnionofPureandAppliedChemistryhaveagreedtoacommonwayofnamingchemicalcompoundsaccordingtoasetofrules,thesenamingsystemistermedIUPACnomenclature.
Therules involved inIUPACnomenclatureareasfollow:
Takeoxygenas–2.
Hydrogenas+1withnon-metals.
Hydrogenas–1withmetals.
Thesenumbersaretheoxidationnumber.
C032-
C+3(-2)=-2
C–6=-2
C=-2+6=+4
Name=trioxocarbonate (IV) ioni.e.3atomsofoxygenandcarbonoxidationnumberis4.
EVALUATION/REVISIONALQUESTIONS
1. Whatarebinarycompounds?
2. Givethecorrectchemicalformulaeof(a)AluminumSulphide,(b)SodiumPhosphate
3. Definethetermaccommodation.
4. Describebrieflywhatismeantby(a)Hypermetropia(b)Myopia
READING ASSIGNMENT
Basis Science Made Easy for JSS Three by F.I Kehinde et al. Chapter 19. Page 68-71.
WEEKEND ASSIGNMENT
- ThesymbolAgrepresentselement(a)Argon(b)Silver(c)Gold(d)Arsenic
- Theelectronicconfiguration2,8,3isthatof________(a)Sodium(b)Aluminum(c)Silicon(d)Argon
- Thecorrectformulaoftetraoxophosphate (V) ionis(a)S042-(b)P033-(c)S032-(d)P043-
- ThecompoundNa3AlF6hastheIUPACname(a)sodiumtrioxoaluminatesIII(b)sodiumhexafloroaluminateIII(c)sodiumheptafloroaluminateIII(d)sodiumtetrafloroaluminateIII
- TheacronymIUPACrepresents(a)InternationalUsageofPureandAppliedChemistry(b)InitiativeUnityforPureandAppliedChemistry(c)InternationalUnionofPureandAppliedChemistry(d)IndoorUnionofplayers,ActorandCorps
THEORY
- Whatarebinarycompounds?
- Stateanytwo examplesofbinarycompounds.
- Writetheelectronicconfigurationof(a)silicon(b)potassium.
- Workouttheoxidationnumberof chromium in Cr2O72-
- What are radicals?
- Statethreeexamples of radicals.
WEEK SEVEN
TOPIC: SIMPLECHEMICALEQUATIONS
CONTENT
- Chemical Equations
- Equationsand Law of Conservation of Matter
- Writing Chemical Equations
- Balancing Chemical Equations
If the law of conservation of matter is tobe kept, itmeansthattheremustbethe same number of atoms in each half of theequation.Theremustbethesamenumberofatomsinreactantsandintheproducts.
Thelawofconservationofmatterstatesthatmatterisneithercreatednordestroyed in the courseofchemicalreaction.This is the mainreasonwhy the number of atomsof productsmustbalancethoseofreactionsand vice versa.
So, the equation:
Na+H2O=Na0H+H2isconsideredunbalanceandhencenotacceptable.
Thishastobebalancedas:
2Na+2H20=2Na0H (aq)+ H2(g)
S=solidL=LiquidsAq=aqueousg–gas
EVALUATION
- Statethelawofconservationofmatter
- Whatisabalanceequation?
WRITINGCHEMICALEQUATIONS
Chemicalequationsarewrittenusingchemicalsymbolsandformulae.It is necessary to have asmuchdetailaspossibleaboutwhat is going oninareactione.g. hydrogen gas reactswithoxygengastoproducewater.
Wetranslatethisinformationtoawordequation.
Hydrogen(gas)+Oxygengas=water
(Reactants) (Product)
 2H2+02 2H20
2H2+02 2H20
Balanced equation
Commonchemicalreactionsinclude(a)Combustionofhydrocarbons(b)Neutralizationreactions (c)Double decomposition
Examples:
 C3H8+02 C02+H20
C3H8+02 C02+H20 HCl+Na0H NaCl+H20
HCl+Na0H NaCl+H20 AgN03+NaCl AgCl+NaN03
AgN03+NaCl AgCl+NaN03
EVALUATION
- Whatkindofreactionisneutralizationreaction?
- Whataretheusualproductsofcombustionofhydrocarbons?
BALANCECHEMICALEQUATIONS
aN2+bH2=cNH3
N:2a =C……(i)
H:2b =3C…..(ii)
Leta=1
Fromequation(i)2(1)=C
2=C C=2
Fromequation (ii)2b=3(2)
2b=6
b=3
A =1, b=3 C=2
N2+3H2=2NH3
Twomethodsarecommonlyemployedinchemicalequations:
- Balancing by inspection.
- Balancing by the use of simultaneous equations.
EVALUATION/REVISIONALQUESTIONS
- Whattwomethodsarecommonlyusedinbalancingchemicalequation?
- Give the balancedequationforthe formation ofwaterfromhydrogenandoxygen.
- What are hormones?
- Whatglandsproducetheminanimals?
WEEKEND ASSIGNMENT
1. Ashortstorywhichtellsuswhathappensinachemicalreactionis known as:(a)chemical reaction(b)chemical formulae(c)chemical formulae(d)chemical balance
2. The subscript (aq)inawrittenchemical equation symbolizes:(a)Aquiline (b) Aquarium(c)Aquarius(d)Aqueous
3. Whatlawgovernsthewrittenofbalancedchemicalequation?(a)lawofconstantcomposition(b)lawofconservationofmatter(c)lawofmultipleproportions(d)law of reciprocal proportions
4. aN2+bH2=CNH3. Whatdoa,b,andcrepresentintheequationwrittenrespectively?
(a) a=3 b=1 c=2(b) a=2 b=3 c=1(c) a=1 b=3 c=2
(d) a=1 b=2 c=3
5. Thereactioninvolvingan acid and abasetoformsaltandwateronlyis known as________(a) Combustion reaction (b)Neutralization reaction (c)Redoxreaction(d)precipitation
THEORY
- Writeoutthechemicalequationofreactiondescribedbelow.(a)Magnesiumribbonreactswith hydrochloricacidtoformmagnesiumchlorideandhydrogengas.
- Whataretheproducts of combustion of hydrocarbons?
WEEK EIGHT
TOPIC: SIMPLEMACHINE
CONTENT
- Definitionof Simple Machine
- Types of Machine
- Importance of Simple Machine
Definition of Simple Machine
A machine is a tool or device that makes work easier. It is defined as a tool which uses force applied (effort) at one point to overcome another resisting force (effort) at another point. Examples of machine include: a plier, the human arm, a gear, a pulley etc.
Types of Machine
There are six types of simple machines. They are;
- Lever
- Wheel and axle
- Pulley
- Wedge
- Inclined plane
- Screw
Lever
A lever is a simple machine built on the principle of a stiff bar resting on a pivot or fulcrum with a load placed at one end and effort applied on the other end.
There are three parts of the lever;
- Load: This is the point of the resisting force.
- Effort: This is the point where force is applied.
- Fulcrum: This is the turning point of the lever.
Types of Lever
There are three classes or order of lever. They are;
 First Order Lever: In this lever, the fulcrum is between the load and effort (LFE). Examples include: scissors, claw hammer.
First Order Lever: In this lever, the fulcrum is between the load and effort (LFE). Examples include: scissors, claw hammer. Second Order Lever: In this lever, the load (L) is between the effort (E) and the fulcrum (F) (FLE). Example of this including; wheelbarrow, nutcracker, bottle opener etc.
Second Order Lever: In this lever, the load (L) is between the effort (E) and the fulcrum (F) (FLE). Example of this including; wheelbarrow, nutcracker, bottle opener etc. Third Order Lever: In this lever, the effort is between load and effort. (FEL). Example of this include; fore arm.
Third Order Lever: In this lever, the effort is between load and effort. (FEL). Example of this include; fore arm.
Wheel and Axle
 This simple machine is used to lift heavy load from depth. It consists of a Large wheel on a small axle that turns in the same axis. Examples of wheel and axle include: bicycles, fan, wagon etc.
This simple machine is used to lift heavy load from depth. It consists of a Large wheel on a small axle that turns in the same axis. Examples of wheel and axle include: bicycles, fan, wagon etc.
Use
- It is used to draw out heavy load from depths
- It is used to create the turning effects in bicycle
EVALUATION
- Define simple machine.
- List two uses of (a) Lever (b)Wheel and Axle.
Pulley
A machine consisting of a wheel over which a pulled rope or chain runs to change the direction of the pull used for lifting a load. Combinations of two or more pulleys working together reduce the force needed to lift a load.
Uses
- It is used lift and move load in cranes
- It is also used in lifts (elevators)
Wedge
A wedge is a triangular shaped tool, and is a portable inclined plane. It can be used to separate two objects or portions of an object, lift up an object, or hold an object in place.
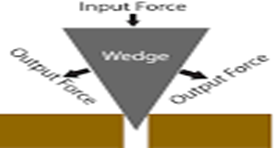
Use
It is used to split objects open especially wood.
Screw
 A screw is a type of fastener characterized by a helical ridge known as thread. Most screw rotate in clockwise manner but few rotate in anticlockwise manner.
A screw is a type of fastener characterized by a helical ridge known as thread. Most screw rotate in clockwise manner but few rotate in anticlockwise manner.
Use
It allows drilling into hard surfaces
Inclined plane
An inclined plane is a flat supporting surface tilted at an angle, with one end higher than the other, used as an aid for raising or lowering a load. The inclined plane is often used to lift a load over a vertical obstacle.

Importance of Simple Machine
- Simple helps man to overcome friction in doing work.
- It helps to move load over a long distance.
- It helps to save time and effort
- It is more efficient.
- Simple machines are basic and simple to operate.
GENERAL EVALUATION\REVISIONAL QUESTIONS
- What is a machine?
- Categorize the following to the class of lever they belong to: fore arm, plier, nutcracker, fishing rod.
- State five effect of radiation.
- State four reasons for abortion.
- What is nuclear transmutation?
READING ASSIGNMENT
Basis Science Made Easy for JSS Three by F.I Kehinde et al. Chapter 17. Pg 70-82.
WEEKEND ASSIGNMENT
- An example of a first order lever is (a) fore arm (b) nutcracker (c) seesaw (d) pulley
- The principle of inclined plane is used in the construction of which of the following (a) pulley (b) staircase (c) lift (d) crane
- ___ has the load between the fulcrum and effort (a) First Order lever (b) Second Order Lever (c) Third Order Lever (d) Wedge
- Which of the following radiation has the lowest penetrating power?(a) alpha rays (b) beta rays (c) gamma rays (d) X-ray
- The clear outer layer of the eye is (a) iris (b) cornea (c) retina (d) lens
THEORY
- Explain the three types of lever giving an example for each.
- State the importance of machine.
WEEK NINE Date:……………………………..
TOPIC: DIGESTIVESYSTEM
CONTENT
- Parts of the Digestive System
- Digestion at Various Parts
- Functions of the Enzymes
- Simple Food Tests
PARTSOFTHEDIGESTIVESYSTEM
The digestive system consists of the alimentary canal, which is a long tube from the mouth, attached to various organs and ends at the anus.
The alimentary canal is a long tube through which food substances pass, become digested (broken down), partly absorbed and the rest passed out.
The alimentary canal has five main parts:(i) The mouth (ii) The gullet (or Oesophagus) (iii) The stomach (iv) The small intestine (v) The large intestine.
 THE HUMAN ALIMENTARY CANAL
THE HUMAN ALIMENTARY CANAL
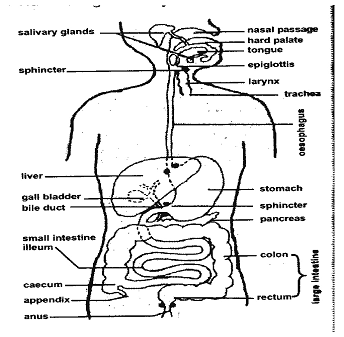
THE MOUTH
It comprises three prominent structures for digestion namely the teeth, salivary glands and the tongue.
TEETH: Structures for food mastication.
SALIVARY GLANDS: Secrete SALIVA (A digestive juice)
TONGUE: Organ of taste.
THE GULLET
It is also known as the Oesophagus. It is lined with smooth involuntary muscles by which contraction of food from the mouth is regulated into the stomach by PERISTALSIS. Peristalsis is the motion by which food is pushed along the gullet and the rest of the digestive tract.
THE STOMACH
This is a muscular organ, which serves in the digestion of food and also acts as temporary food store. A digestive juice called the GASTRIC JUICE is secreted into the stomach by both the PEPTIC and OXYNTIC CELLS.
THE SMALL INTESTINE
The longest portion of the digestive tract (about 9m in length if fully stretched out) which begins with the C-shaped DUODENUM on which the LIVER and PANCREAS are attached. Both discharge their secretions – THE BILE and the PANCREATIC JUICE respectively to the duodenum.
THE LARGE INTESTINE
Also known as COLON. It processes undigested food portion as FAECES for export outside the body through the ANUS. No digestion occurs in it. Excess water is also removed through the feaces by large intestine.
EVALUATION
1. Mention the five main parts of the human digestive system.
2. What digestive juices are present along the digestive tract?
DIGESTION AT VARIOUS PARTS
A balanced diet consists of carbohydrates, fats & oils, proteins, mineral salts, vitamins and water. The carbohydrates, proteins and fats are large complex molecules. They must be broken down into small simpler molecules before the body can use them.
The conversion of large complex molecules to small simpler molecules is the process of digestion.
Digestion is defined as the breakdown of large complex molecules in foods into small simpler absorbable molecules by the enzymes. Digestion takes place in the mouth, stomach and the small intestine. There are four digestive juices along the digestive tract. These are:
(i) SALIVA
(ii) GASTRIC JUICE
(iii) PANCREATIC JUICE
(IV) INTESTNAL JUICE
Each of them contains one or more digestive enzymes, which converts specific food complex to simpler forms.
The following are the components of each digestive juice.
Parts of Alimentary Canal | Digestive Juice | Components |
Mouth | Saliva | Ptyalin + Water |
Stomach | Gastric juice | Pepsin + Rennin |
S.I (Duodenum) | Pancreatic Juice | Amylase + Lactase + Sucrase + Trypsin |
S.I (Ileum) | Intestinal Juice | Maltase + Lactase + Sucrase + Erepsin + Lipase |
FUNCTIONS OF THE ENZYMES
Ptyalin – converts cooked starch to maltose.
Pepsin – converts protein to peptones and polypeptides.
Rennin- coagulatesliquid proteins (milk) into thick digestive form.
Trypsin – breaks down protein to peptones and polypeptides.
Amylase – breaks down starch into maltose.
Lipase – breaks down fats into fatty acid and glycerol.
Maltase – breaks maltose to glucose.
Lactase – breaks lactose to glucose & fructose.
Sucrase – breaks sucrose to glucose &galactose.
Erepsin – breaks polypeptides into amino acid.
Mouth: Digestion of starch begins in the mouth. Ptyalin converts starch to maltose sugar in the mouth.
Stomach: Only proteins are digested in the stomach. Pepsin converts solid proteins into peptones and polypeptides while Rennin curdles milk proteins.
Small Intestine (Duodenum): The three major classes of foods are all digested here.
Trypsin – converts protein to peptones and polypeptides.
Amylopsin – converts starch to maltose.
Lipase – converts fats& oil to fatty acid & glycerol.
Small Intestine (Ileum) – Digestion is completed here as all partially digested food substance are completely digested.
Maltase – breaks down maltose to glucose.
Erepsin – breaks down polypeptides to amino acid.
Lipase – breaks down fats & oil to fatty acid and glycerol.
These final products of digestion in the Ileum are the small simple molecules which can be absorbed into the body.
ABSORPTION OF FOOD
When food has been finally digested to simple absorbable forms, they are simply absorbed in the small intestine into the blood stream by diffusion through several fingers like projections on the internal surface area of the ileum.
These projections are known as VILLI. Each villus comprises a network of capillaries and the lateal vessels. Glucose and Amino acid diffuse into the network of capillaries of villi into the blood stream while the lateal vessel takes up fatty acid and glycerol.
Indigestible portion of the food is regulated into the larger intestine where it is dehydrated and processed into semi solid feaces for egestion.
EVALUATION
1. a. Name the components of a balanced diet.
b. State which components of the balanced diets are large complex molecules that require digestion?
2 a.Name the four digestive juices.
b.Mention the enzymes present in each juice.
3. Explain the following (a) absorption of food (b) egestion.
SIMPLE FOOD TESTS
1. Test for starch
Starch + Iodine solutions
Result: Blue – black colour confirms the presence of starch
2. Test for reducing sugars e.g. glucose
Glucose + Fehling solutions
Boil the mixture
Result: Brick – red precipitate confirms the presence of reducing sugar
3. Test for Non-reducing sugars eg Sucrose
Sucrose + Dill Hcl acid
Boil for about 5 minutes
Cool the mixtures
Add some NaHCO3 until there is fitting
Now add Fehling’s solution
Warm the tubes
Result: Brick – red precipitate obtained
N.B: If Sucrose is not first heated as described, it would give no colour with Fehling solution for it is a non-reducing sugar. The HCl breaks down the sucrose into simpler sugar, which reacts with the Fehling solution.
4. Test for protein
2cm3 of protein (milk) + 2-3 drops of Million’s reagent
Boil the mixture
Result: Orange colour or Brick red precipitate confirms presence of protein.`
5. Test for lipids
oil + Sudan III solution
Result: Red stains confirm presence of oil or fat
OR: oil + paper
Result: Paper turns translucent to show that lipid is present
6. Test for water
Water + white anhydrous CuSO4
Result: Blue colour confirms presence of water
OR: Water Blue cobalt II chloride paper
Result: Pink colour confirm the presence of water
GENERALEVALUATION
- Classify the following sugars either as reducing or non-reducing (a) Sucrose (b) Maltose. (c) Glucose (d) Lactose (e) Galactose (f) Fructose.
- Mention the reagent needed to test for each of the following: (a) Starch (b) protein (c) reducing sugar (d) Lipid (e) water (f) non-reducing sugar
- (a) Name the components of a balanced diet (b) State which components of the balanced diets are large complexmolecules that require digestion?
- (a)Name the four digestive juices. (b) Mention the enzymes present in each juice.
- Explain the following (a) absorption of food (b) egestion.
READING ASSIGNMENT
Integrated Science Made Easy, bk two by F.I. Kehinde. Pgs 13-15.
WEEKEND ASSIGNMENT
1. The process by which complex molecules in food are reduced into simpler absorbable one is
A. Egestion B. ingestion C. digestion D. absorption
2. One of these is NOT a digestive enzymeA. Mucin B. ptyalin C. pepsin D. amylase
3. Which reagent is most suitable for testing for presence of starch in heat? A. Osmic acid B. iodine C. million’s reagent D. Sudan III solution.
4. All of these are digestive juices except A. Salivary glands B. pancreatic juice C. Bile pigmentD. succusentericus
5. Oxyntic cells secretes ………… into the stomach A. Dilute HCl B.Trypsin C. pepsin D. rennin
THEORY
- Mention the five main parts of the digestive system.
- Define the following (a) peristalsis (b) egestion.
WEEK TEN Date:……………………………..
TOPIC: THE CIRCULATORY SYSTEM
CONTENT
- Blood Components and Functions.
- The Heart and Blood Vessels.
- The Human Circulatory System
- Blood Defects and Diseases
Blood Components and Functions
Blood is a liquid connective tissue, which is maintained in constant circulation round the body. In certain organisms, blood flows within a closed system of inter connecting vessels while the reverse holds in other organisms.
The human blood is of PH 7.4 and comprises four components:
1. Plasma [liquid part of blood]
2. Red Blood cells [Erythrocytes]
3. White Blood Cells [Leucocytes]
4. Blood platelets [Thrombocytes]
PLASMA
This is the liquid part of blood, which makes up about 55% volume of blood. The plasma itself is made of about 90% water, proteins and mineral salts. It takes digested food from the small intestine to places where needed and also takes waste product to the excretory organs for removal.
RED BLOOD CELLS: These are the most numerous cells in the blood. They account for the redness of blood. Each red cell contains HEAMOLOGIN, a respiratory pigment which enables Red Blood Cells to transport oxygen in the blood. Red blood cells are also called ERYTHROCYTES. They are formed in the BONE MARROW and destroyed in the LIVER and SPLEEN when too old to perform their functions completely after about 120days of existence.
WHITE BLOOD CELLS
They are the least numerous cells in the blood. They fight microorganisms and infections in the blood. WBC also helps the body to destroy bacteria and viruses through the agency of ANTIBODIES in the blood plasma.
PLATELETS
The second most numerous blood cells which help blood to clot in wounds. They are also called THROMBOCYTES. They are tiny cells, which are much smaller than either the RBCs or WBCs. Most often they are found as fragments.
EVALUATION
1. What is Blood?
2. State the components of blood.
3. Explain briefly each blood component.
The Heart and Blood vessels
The heart is the muscular pumping organ, which maintains a continual flow of blood round the body. The human heart comprises four chambers namely:
a. The 2 upper and smaller chambers [ARTRIA or AURICLES]
b. The 2 Lower and bigger chambers called VENTRICLES
The heart is connected to blood vessels within which blood is transported round the body.
THE BLOOD VESSELS

Three types of blood vessels known are:
Veins : Return blood to the heart
Arteries: Carry blood away from the heart.
Capillaries: Link arteries with venules
 Differences between Arteries and Vein
Differences between Arteries and Vein
 ARTERIES VEINS
ARTERIES VEINS
1. Carry blood away from the heart Returns blood to the heart
2. Carry oxygenated blood at high Carry deoxygenated blood under low pressure pressure
3. Are deeply located under the skin. Are superficially located beneath the skin
EVALUATION
1. Mention the chambers associated with blood vessels and valves of the human heart.
2. State two differences between arteries and veins.
THE HUMAN CIRCULATORY SYSTEM
It is described as a closed circulatory system in that blood is circulated round the body through a system of interconnecting blood vessels. The human circulatory system is also described as double circulation in that blood has to flow twice through the heart before one complete circulation can be made through each time it does. It flows through separate pathways.
The two pathways are known as the:
a. Lung or Pulmonary pathway.
b. Body or systemic pathway.
The lung pathway ensures re-oxygenation of the blood while the body pathway ensures delivery of oxygen to the tissues and cells of the body.
EVALUATION
- What does it mean to describe the human circulatory system as:a. Closed circulationb. Double circulation?
- What kind of blood is conveyed by the a. Pulmonary artery b. Pulmonary vein c. Aorta d. Vena cava?
Blood Defects and Diseases
Some people may suffer through not having enough RBCs and hence are unable to get sufficient supply of oxygen.These people can be said to be Anaemic.Thus, anemia is a serious blood defect. Other blood diseases abound and are named below viz:
1. Leukaemia
2. Haemophilia
3. Sickle cell
4. Septicemia
5. Hypertension
6. Hypotension
Leukaemia: Imbalance of red and white blood cells as there is a preponderance of defective and cancerous WBCs. This is caused by exposure to radioactive fall-out from atomic bombs or by over exposure to x-rays.
Sickle Cell: This is a disease which arises from the formation of sickle shaped red blood cells in the blood and which are unable to deliver the oxygen requirement of the body promptly and effectively. It is hereditary.
Haemophilia: Inability of the blood to check blood loss automatically when minor injuries are sustained. It is also hereditary.
Septicemia: Blood poisoning due to certain bacterial infections and which results in lacking of the blood
Hypertension: Describes the clinical condition of high blood pressure.
Hypotension: Low blood pressure. Occasionally, bleeding may occur when blood vessels are damaged or ruptured in accidents, wounds and certain illnesses.
GENERAL EVALUATION
1 Mention two heritable blood diseases.
2 Distinguish between hypertension and hypotension.
3. What is Blood?
4. State the components of blood.
5. Explain briefly each blood component.
READING ASSIGNMENT
Integrated Science Made Easy, bk two pgs 21-25
WEEKEND ASSIGNMENT
- Which one of these is NOT a Blood compound cell?A. Plasma B. Erythrocytes C. Leucocytes
D.Thrombocytes
- A disease caused by a preponderance of cancerous and defective white blood cells in the blood describes A. Septicemia B. Leukemia C. Hemophilia D. Hypertension
- The blood vessels, which look arterioles up with vennules, are known as A. Arterioles B. Vena cava C. Capillaries D. Vennules
- The chamber of the heart with the thickest wall is the A. Left ventricle B. right auricle C. left auricle D. right ventricle
- The largest artery in the body is the A. Pulmonary artery B. aorta C. hepatic artery D. renal artery
THEORY
- Draw and label the human heart.
- Write briefly on the human circulatory system.











