HEAT-2
UNIT OF R
From, R = = K
From R = …
Hence the SI unit used is Kelvin per watt (K).
DETERMINATION OF THERMAL CONDUCTIVITY OF A GOOD CONDUCTOR OF HEAT
SEARLE’S METHOD
The heater is switched on and water is passed through the copper coil at a constant rate.
If the bar is assumed to be perfectly lagged, then at steady state (i.e., when all four thermometers give steady readings) the rate of flow of heat in distance X is given by:
……………… (1)
Where K = Thermal conductivity of the material of the bar.
Since the bar is assumed to be perfectly lagged, all of the heat which flows along the bar is used to increase the temperature of the water.
……………………….. (2)
Where m = mass of water flowing per unit time.
C = specific heat capacity of water.
Equation (1) = Equation (2)
―K‖ can be found from this equation.
The holes at X contain oil or mercury to ensure good thermal contact between the thermometers and the bar.
DETERMINATION OF THERMAL CONDUCTIVITY OF A BAD CONDUCTOR OF HEAT
LEES’ DISC METHOD
The thermal conductivity of a bad conductor of heat such as glass, asbestos wool, paper, etc., can be determined by using Lees’ disc method.
With this method, a bad conductor of heat such as asbestos wool is sandwiched between a steam chest C and a thick brass slab B.
The arrangement is suspended on three strings which are attached to B. Thermometers are placed as shown, and to ensure good thermal contact, some mercury or Vaseline is applied in the holes where thermometers are inserted.
The lower face of C and the upper face of B are made flat for uniform heat flow.
Steam is passed through the chest and the apparatus is left to reach steady state.
At steady state we have:
Where K = Thermal conductivity of the sample, A = Area of one face of the sample, l = Thickness of the sample.
Here the rate at which heat is flowing through the sample is equal to the rate at which B is losing heat to the surroundings.
Now the sample is removed so that B comes into direct contact with C and is heated by it.
When the temperature of B has risen by about 10°C, C is removed and the sample is replaced on top of B.
Since C is no longer present, B cools and its temperature is recorded at one-minute intervals until it has dropped to about 10°C below its steady state temperature.
A graph of temperature against time is plotted.
Slope of tangent at = …
Where = mass of brass slab B
Specific heat capacity of brass slab B
The conditions under which B is losing heat are the same as those at steady state and hence
Equation (1) = Equation (2)
Thus K can be determined from this equation.
Problems
- What does it mean by thermal conductivity of a substance?
- Find the heat lost per square meter through a cavity wall when the temperature difference between the inside and outside is 15°C, given that each of the two brick layers is 100mm thick and the cavity is also 100mm across.
Brick = 1.0 Wm-1K-1
Air = 0.025 Wm-1K-1
Problem 16
Calculate the quantity of heat conducted through 2m2 of brick-wall 12cm thick in 1 hour if the temperature on one side is 8°C and the other side is 28°C. Given that thermal conductivity of brick = 0.13 Wm-1K-1.
Problem 17
Estimate the rate at which ice melts in a wooden box 2cm thick and inside measurements 60cm x 60cm x 60cm, assume that external temperature is 27°C and coefficient of thermal conductivity of wood = 0.1674 Wm-1K-1. Specific latent heat of fusion of ice = 336 x 103 Jkg-1.
Problem 19
Two cylinders of equal physical dimensions are placed one on top of the other as illustrated below.
The lower surface of the silver cylinder is kept at 0°C and the upper surface of the iron cylinder is kept at 100°C. Given that the thermal conductivity of silver is eleven times that of iron, calculate the temperature of the surface AB.
Problem 20
A composite bar is made of a bar of copper 10cm long, a bar of iron 8cm long, and a bar of aluminium 12cm long, all having the same cross-sectional area. If the extreme ends of the bars are maintained at 100°C and 10°C respectively, find the temperature at the two junctions. Given that thermal conductivity of copper, iron, and aluminium are 400, 40, and 20 Wm-1K-1 respectively.
Problem 21
A window of height 1.0m and width 1.5m contains a double glazed unit consisting of two single glass panes, each of thickness 4.0mm separated by an air gap of 70mm. Calculate the heat energy per second conducted through the window when the temperature difference across the unit is 10K.
Problem 22
An electric heater is used in a room of total wall area 137m2 to maintain a temperature of 20°C inside it when the outside temperature is 10°C. The walls have three layers of different materials. The innermost layer is of wood of thickness 2.5cm, the middle layer is of cement of thickness 1.0cm, and the outermost layer is of brick of thickness 25cm. Find the power of electric heater. Assume that there is no heat loss through the floor and ceiling. Thermal conductivity of wood, cement, and brick are 1.25, 1.5, and 1.0 Wm-1K-1 respectively.
Problem 23
- What does it mean by thermal conductivity of a substance?
- Find the heat lost per square meter through a cavity wall when the temperature difference between the inside and outside is 15°C, given that each of the two brick layers is 100mm thick and the cavity is also 100mm across.
Problem 24
- Assuming you are managing a metal box company, what requirements for thermal conductivity, specific heat capacity, and coefficient of expansion would you want a material to be used as a cooking utensil to satisfy?
- A hot water boiler consists of iron wall of thickness 2.0cm and effective inner area of 2.5m2. The boiler is heated by a furnace and generates high pressure steam of temperature 170°C at the rate of 1.2 kg/min. The latent heat of steam at 170°C is 2.09 x 106 J/kg. Assuming the outer face of the boiler to be at a temperature of 178°C, what is the coefficient of thermal conductivity of iron?
Problem 25
A copper kettle has a circular base of radius 10cm and thickness 3.0mm. The upper surface of the base is covered by a uniform layer of scale 1.0mm thick. The kettle contains water which is brought to the boil over an electric heater. In the steady state condition, 5.0g of steam is produced each minute. Determine the temperature of the lower surface of the base assuming the condition of heat along the surface of the kettle can be neglected. Thermal conductivities:
- Copper: 3.8 x 102 Wm-1K-1
- Scale: 1.34 Wm-1K-1
Specific latent heat of steam = 226 x 103 J/kg.
Problem 26
- Define thermal conductivity of a material.
- Heat is supplied at the rate of 80W to one end of a well-lagged copper bar of uniform cross-sectional area 10cm2 having a total length of 20cm. The heat is removed by water cooling at the other end of the bar. Temperature recorded by two thermometers at distances 5cm and 15cm from the hot end are 48°C and 28°C respectively.
- Calculate the thermal conductivity of copper.
- Estimate the rate of flow (in g/min) of cooling water sufficient for the water temperature to rise by 5K.
- What is the temperature of the cold end of the bar?
U – VALUE / THERMAL TRANSMITTANCE
The U-value of a structure (cavity wall or a window) is the heat transferred per unit time through unit area of the structure when there is unit temperature difference across it.
U-value =
This can be obtained from:
or
U-values provide architects and building engineers with information about heat losses from buildings.
They take account not only of heat lost by conduction, but of any lost by convection or radiation.
Architects base their calculations on U-values rather than on coefficients of thermal conductivity because they are concerned with the air temperature inside and outside a room, not with the temperature on the surfaces of a piece of glass, a wall, etc.
THERMAL CONVECTION
This is the process by which heat is transferred from one part of a fluid to another by the movement of the fluid itself.
Here the molecules of a fluid (liquid/gas) carry heat energy from one part of a fluid to another.
TYPES OF CONVECTION
- Natural / free convection
- Forced convection
NATURAL FREE CONVECTION
This is a type of convection in which a heated fluid flows from the hot region to the cold region due to differences in density.
Example: When a fluid is heated from below, the lower part of the fluid becomes hot and therefore expands.
Its density decreases due to the increase in volume of fluid molecules. Its position is displaced by cold fluid from the top. This in turn gets heated and rises to the top and this process continues as shown in the figure below.
FORCED CONVECTION
This is a type of thermal convection in which the heated fluid is forced to move from the hot region to the cold region by means of a blower or pump, i.e., by an external agent.
GOVERNING LAWS
Thermal convection is governed by two laws:
- Newton‘s law of cooling
- Dulong and Petit law
NEWTON’S LAW OF COOLING
Newton‘s law of cooling is approximately true in still air only for a temperature excess of about 20K or 30K; but it is true for all excess temperature in conditions of forced convection of the air, i.e., in draught.
Statement of the law: The rate of loss of heat to the surrounding air is proportional to the excess temperature over the surroundings.
Excess temperature: The excess temperature of a body over the surrounding is the difference between the temperature of the body and that of the surrounding.
Let T be temperature of the body and T₀ be temperature of the surrounding.
Excess temperature = T – T₀
Let R be rate at which a body is losing heat.
From Newton‘s law of cooling:
Where K = convection coefficient.
Negative sign shows that the body is losing heat.
If m is the mass of a body and C is its specific heat capacity, then
For a given body mc = constant.
Using (2) in (1) we have:
DULONG AND PETIT LAW
Dulong and Petit modified Newton‘s law of cooling and stated a law which works under natural/free convection.
Condition of the law: The law works under natural/free convection where θ – θₛ > 50K.
Where the law works: The law works in still air in the laboratory.
Alternative name of the law: Five-fourth power law.
Statement of the law: Under natural/free convection, the rate at which a body loses heat is proportional to the fifth power of its excess temperature over the surroundings.
Where K = Convectional coefficient.
Negative sign shows that the body is losing heat.
Problem 28
A body cools from 40°C in 5 minutes. The temperature of the room being 15°C, what will be the temperature of the body after another 5 minutes?
Problem 29
In a room at 15°C, a body cools from 35°C in 4 minutes. Find the further time elapsed before the temperature of the body is 20°C.
Problem 30
Wind blows over a hot liquid placed in a beaker in the laboratory whose average room temperature is 27°C. The liquid rate of cooling is 15°C/min when it is at a temperature of 87°C. Calculate the liquid rate of cooling when it is at a temperature of 57°C.
Problem 31
A body initially at 80°C cools to 64°C in 5 minutes and 52°C in 10 minutes. What will be the temperature after 15 minutes and what is the temperature of the surrounding?
THERMAL RADIATION
This is the transfer of heat energy from one point to another without the requirement of any material medium.
It is like a throw of radiant energy.
Thermal radiation consists of electromagnetic waves with a range of wavelengths covering the infra-red and visible regions of the electromagnetic spectrum.
All bodies continuously emit and absorb thermal radiation in the form of electromagnetic waves. A body at higher temperature than the surrounding emits more radiation than it absorbs.
Thus, there is a continuous exchange of radiation between the body and the surrounding with the result that there will be a rise or fall in temperature of the body.
THE BLACK BODY
A perfectly black body is one which absorbs completely all the radiation falling on it and reflects none.
Since a perfectly black body is a perfect absorber, it will also be a perfect radiator.
When a perfectly black body is heated to a high temperature, it emits thermal radiation of all possible wavelengths.
Practical examples of perfectly black bodies:
- The sun
- A surface coated with lamp-black
This surface can absorb 96% to 98% of the incident radiation and may be considered as a perfect black body for all practical purposes.
HOW TO REALIZE A BLACK BODY
A good black body can be realized simply by punching a small hole in the lid of a closed empty tin.
The hole looks almost black, although the shining tin is a good reflector.
Reason: The hole looks almost black because the radiation that enters through it is reflected from the inside walls several times and is partially absorbed at each reflection and loses energy until no radiation is reflected back. Hence the hole absorbs all radiation falling on it.
BLACK BODY RADIATION (BBR)
Is the thermal radiation emitted by a black body at a given temperature.
Any object at a temperature greater than absolute zero emits thermal radiation of all wavelengths within a certain range.
The amount of thermal energy radiated for different wavelength intervals is different and depends on temperature and nature of the surface.
INTENSITY OF RADIATION
Symbol I
This is the rate at which radiant energy is transferred per unit area.
I =
The SI unit used is Watt per square meter (Wm-2).
LAWS OF BLACK BODY RADIATION
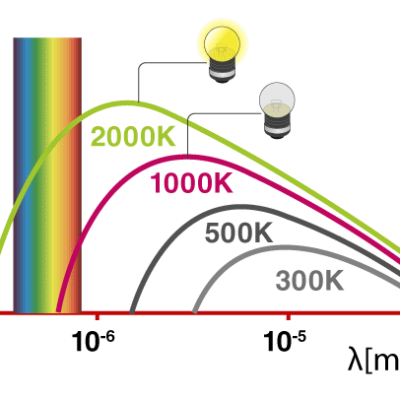
Law 1: WIEN’S DISPLACEMENT LAW
The wavelength λ at which the maximum amount of energy is radiated by a black body is inversely proportional to its absolute temperature.
Where K = constant of proportionality called Wien‘s constant (k) whose value is 2.9 x 10-3 m·K.
λmax T = K
Law 2: STEFAN’S LAW / STEFAN–BOLTZMANN’S LAW
The total energy emitted per unit area of a black body in unit time is directly proportional to the fourth power of its absolute temperature.
E =
Where K = constant of proportionality called Stefan‘s constant (σ) whose value is 5.67 x 10-8 Wm-2K-4.
Problem 33
Assuming the total surface area of the human body is 1.25m2 and the surface temperature is 30°C, find the total rate of radiation of energy from the human body. Given that Stefan’s constant, σ = 5.67 x 10-8 Wm-2K-4.
Problem 34
A black ball of radius 1m is maintained at a temperature of 30°C. How much heat is radiated by the ball in 4 seconds? Given that Stefan’s constant = 5.67 x 10-8 Wm-2K-4.
Problem 35
Two spheres made of the same material have radii 2.0cm and 3.0cm and their temperatures are 627°C and 527°C respectively. If they are black bodies, compare:
- The rate at which they are losing heat.
- The rate at which their temperatures are falling.
EMISSIVITY (ε)
The emissivity (ε) of a surface is the ratio of the power radiated by a surface of a given body to that radiated by a black body at the same temperature.
From Stefan’s law:
P = εσAT4
Where P is power radiated by surface of given body.
Problem 36
A tungsten filament of total surface area of 0.45 km2 is maintained at a steady temperature of 2227°C. Calculate the electrical energy dissipated per second if all this energy is radiated to the surrounding.
Given that emissivity of tungsten at 2227°C = 0.3 and Stefan’s constant = 5.67 x 10-8 Wm-2K-4.
Problem 37
The tungsten filament of an electric lamp has a length of 0.5m and a diameter of 6 x 10-5m. The power rating of the lamp is 60W. Assuming the radiation from the filament is equivalent to 80% that of a perfect black body radiator at the same temperature, estimate the steady temperature of the filament given that Stefan’s constant = 5.67 x 10-8 Wm-2K-4.
T = 935.5K
PROVOST’S THEORY OF HEAT EXCHANGE
A body radiates heat at a rate which depends only on its surface and temperature, and it absorbs heat at a rate depending on its surface and the temperature of its surroundings.
THEORY
When the temperature of a body is constant, the body is losing heat by radiation and gaining it by absorption at equal rates.
Consider a black body at a temperature T placed in an enclosure having temperature T₀.
Energy radiated /sec = A T4
Energy absorbed /sec = A T₀4
If the body is not at the same temperature as its surrounding, there is a net flow of energy between the surrounding and the body because of unequal emission and absorption.
If the temperature of the body is greater than that of the surrounding, then the net energy will flow from the body to the surrounding.
Net energy emitted /sec = A (T4 – T₀4)
If the temperature of the body is less than that of its surrounding, then the energy will flow from the surrounding to the body.
Net energy absorbed /sec = A (T₀4 – T4)
Problem 38
The total external surface area of a dog’s body is 0.8 m2 and the body temperature is 37°C. At what rate is it losing heat by radiation when it is in a room whose temperature is 17°C? Assume that dog’s body behaves as a black body and given that Stefan’s constant is 5.67 x 10-8 Wm-2K-4. [98.086W]
Problem 39
The cathode of a certain diode valve consists of a cylinder 2 x 10-2 m long and 0.1 x 10-2 m in diameter. It is surrounded by a co-axial anode of diameter larger than that of the cathode. The anode remains at a rate temperature of 127°C when the power of 4 watts is dissipated in heating the cathode.
Estimating the temperature of cathode. [1035.29]
List the assumptions you have made in arriving at your estimate.
Problem 40
A metal sphere with a black surface and radius 30 mm is cooled to 200K and placed inside an enclosure at a temperature of 300K. Calculate the initial rate of temperature rise of the sphere, assuming the sphere is a black body. Given that:
- Density of metal = 8000 kg/m3
- Specific heat capacity of metal = 400 J/kg·K
- Stefan’s constant = 5.67 x 10-8 Wm-2K-4
Problem 41
The energy arriving per unit area on the earth’s surface per second from the sun is 1.34 x 103 Wm-2. The average distance from the earth to the sun is 215 times as great as the sun’s radius. Given that both the earth and the sun are black bodies.
Stefan’s constant = 5.67 x 10-8 Wm-2K-4. [T = 575K]
Problem 42
The amount of radiant heat received by the earth from the sun is 1.34 x 103 Wm-2. Suppose all these radiations on the earth are re-emitted by the earth. Calculate the temperature of the earth. Given Stefan’s constant = 5.67 x 10-8 Wm-2K-4. [T = 395K]
Problem 43
The surface temperature of the sun is 6000K. If we consider it as a perfect black body, calculate the energy radiated by the sun per second.
Given that the radius of the sun = 6.92 x 108 m and Stefan’s constant = 5.67 x 10-8 Wm-2K-4. [P = 4.42 x 1026 W]
Problem 44
The temperature of a furnace is 2324°C and the intensity is maximum in its radiation spectrum nearly at 12000Å. If the intensity in the spectrum of a star is maximum nearly at 4800Å, then calculate the surface temperature of the star. [T = 6492.5K]
Problem 45
The wavelength corresponding to maximum energy for the moon is 14 x 10-6 m. Estimate the temperature of the moon if b = 2.884 x 10-3. [T = 206K]
ALBEDO
Each planet absorbs a certain fraction of energy from the sun and the rest of the energy is reflected back into space.
Definition: Albedo is the ratio of the sun’s energy reflected by a planet to the energy falling on it.
IMPORTANCE OF ALBEDO
It helps to know whether a certain planet has a cloud cover or not.
Clouds are good reflectors of radiant energy and hence increase the reflecting power of a planet.
High value of albedo means the planet has dense clouds.
GAS LAWS
Gas laws try to describe the behavior of gases in relation to temperature, pressure, and volume.
A gas law is obtained when two of these three quantities vary while the third is kept constant.
BOYLE’S LAW
The law states that “The pressure P of a given mass of a gas is inversely proportional to its volume V at constant temperature”.
The relationship can be represented graphically as shown below.
The graph shows that when the volume is increased from V1 to V2, the pressure decreases from P1 to P2 and vice versa.
CHARLES’ LAW
The volume of a fixed mass of gas at constant pressure is proportional to its thermodynamic temperature.
Where K = constant of proportionality.
IDEAL GAS [PERFECT GAS]
An ideal gas is a hypothetical gas that obeys gas laws exactly.
It consists of molecules that occupy negligible volume of the amount n of a gas.
Boyle’s law and Charles’ law can be combined together to give a single equation which describes the temperature, pressure, and volume behavior of the gas.
Boyle’s law: V ∝ 1/P (at constant T)
Charles’s law: V ∝ T (at constant P)
Combined: PV = nRT
Where R = universal gas constant.
NUMERICAL VALUE OF R
At S.T.P:
Summary
- Thermal conductivity is the ability of a material to conduct heat.
- U-value measures heat transfer through a structure per unit area per unit temperature difference.
- Thermal convection transfers heat by fluid movement; natural convection occurs due to density differences, forced convection by external means.
- Newton’s law of cooling and Dulong and Petit law describe heat loss by convection.
- Thermal radiation transfers heat via electromagnetic waves; black bodies absorb and emit radiation perfectly.
- Wien’s displacement law relates peak wavelength to temperature.
- Stefan-Boltzmann law relates total radiation emitted to the fourth power of temperature.
- Emissivity measures how well a surface emits radiation compared to a black body.
- Gas laws describe relationships between pressure, volume, and temperature of gases.




12 Comments