Carbon and its compounds Questions
1. (a) State one use of graphite
(b) Both graphite and diamond are allotropes of element Carbon. Graphite conducts electricity
whereas diamond does not. Explain
2. Below is a simplified scheme of solvay process. Study it and answer the questions that follow:
a) Identify gas R.
b) Write an equation for the process III.
c) Give one use of sodium carbonate.
3. A burning magnesium continues to burn inside a gas jar full of carbon (IV) oxide. Explain.
4. The diagram below shows a jiko when in use

(a) Identify the gas formed at region H
(b) State and explain the observation made at region G
5. Study the diagram below and use it to answer the questions that follow.
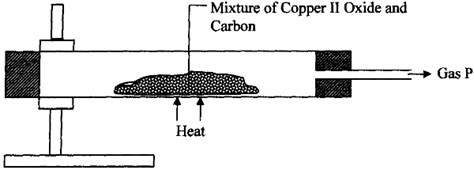
(a) State the observation made in the combustion tube.
(b) Write an equation for the reaction that took place in the combustion tube
(c) Give one use of P
6. (a) Identify two substance that are reacted to regenerate ammonia gas in the solvary process
(b) Write down a balanced chemical equation for the reaction above
7. When the oxide of element H was heated with powdered Carbon, the mixture glowed and
Carbon (IV) oxide was formed. When the experiment was repeated using the oxide of element J,
there was no apparent reaction
(a) Suggest one method that can be used to extract element J from its oxide
(b) Arrange the elements H, J and Carbon in order of their decreasing reactivity
8. (i) Diamond and silicon (IV) Oxide have a certain similarity in terms of structure and bonding.
State it
(ii) State one use of diamond
9. (a) What is allotropy?
(b) Diamond and graphite are allotropes of Carbon. In terms of structure and bonding
explain why graphite conducts electricity but not diamond
10. The diagram below shows a charcoal stove with different regions
(a) Write an equation for the formation of the product in region B
(b) How would one avoid the production of the product at B? Give a reason for your answer
- Study the diagram below and answer the questions that follow:
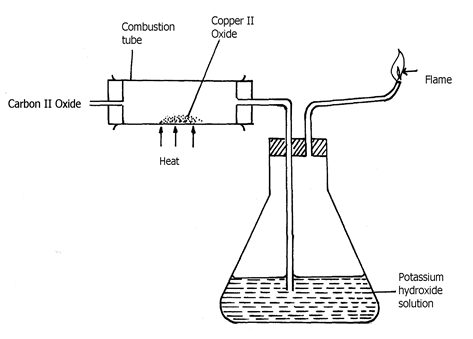
(a) Explain the observation made in the combustion tube during the experiment
(b) Write an equation for the reaction that takes place in the combustion tube
12. Diamond and graphite are allotropes of carbon:-
(a) What is meant by allotropes?
(b) How do they differ in their structure and bonding
13. Study the experimental set-up below:
a) State two observations made in the set up as the experiment progressed
b) By use of a chemical equation, explain the changes that occurred in the boiling tube
c) Why was it necessary to burn the excess gas?
14. The diagram below shows the heating curve of a pure substance. Study it and answer the
questions that follow:
(a) What physical changes are taking place at H and W?
(b) What are the physical states of the substance at Y and K?
(c) Using the simple kinetic theory of matter, explain what happens to the substance between
points A and C
(d) The substance under test is definitely not water; Give a reason for this
(e) What would happen to the melting point of this substance if it were contaminated
with sodium chloride?
(f) What happens to the temperature between points B and C?
15. Study the set-up below and answer the questions that follow:

(a) (i) Name Gas X ………………………………………………………………
(ii) State the effect of releasing gas X to the environment
(b) Write down equations for the reactions taking place in;
(i) Tube I
(ii) Tube II
(iii) Flask
(c) State the observation made in tube III
(d) Write down an equation for the reaction which could be used to generate Carbon
(IV) Oxide for the above set up
(e) Name the reagents used to generate gas x in the laboratory
(f) Complete the diagram above to show how excess gas x can be collected
16. The figure below shows the stages in the manufacture of sodium carbonate. Study the diagram
below and use it to answer the questions that follow.
a) (i) Name three starting materials in the manufacturer of sodium carbonate.
(ii) Which substances are recycled in this process?
(iii) Identify the chambers in which the recycled substances are regenerated.
(iv) Name the substances U and V.
b) Give an equation for the reaction which occurs:
(i) In the reaction chamber 1
(ii) When solid V is heated.
(iii) In the reaction chamber 3.
c) State one commercial use for
(i) Sodium carbonate.
17. The set-up below was used to prepare dry carbon (II) Oxide gas. use it to answer the questions
below it:
(a) (i) State two mistakes committed in the set-up arrangement above
(ii) The student produced carbon (IV) oxide gas from the reaction between Lead (II) Carbonate
and dilute hydrochloric acid. The gas was produced for a short time and the reaction came
to a stop. Explain
(iii) Write the equation for the reactions taking place in the combustion tube and the conical
flask:
Combustion tube:…………………………………………………………………..
Conical flask ……………………………………………………………………..
(iv) State one use of carbon (IV) Oxide gas apart from fire extinguisher
(v) Give two properties that make carbon (IV) Oxide to be used as fire extinguisher
(b) PbO(s) + CO(g) Pb(s) + CO2(g)
Which property of carbon (II) Oxide is demonstrated by the above equation?
(c) Aluminium carbonate does not exist. Give a reason
(d) Ammonium carbonate decomposes when heated. Write a chemical equation to
represent this decomposition
18. State and explain the observation made when a piece of charcoal is dropped in a jar containing
concentrated nitric (V) acid
19. When Carbon (IV) oxide is passed through lime water, a white precipitate is formed but
when excess Carbon (IV) Oxide is passed, the white precipitate disappears;
(a) Explain why the white precipitate disappears
(b) Give an equation for the reaction that takes place in (a) above
20. The set-up below was used to prepare a carbon (II) oxide gas.
(a) Give the name of substance A ………………………………………………………….
(b) Complete the diagram to show how the gas can be collected
(c)Write the equation for the reaction
6. Carbon and its compounds Answers
1. a) – making of pencil
– As a lubricant
b) Graphite has delocalized in its structure hence it conducts electricity. Carbon uses all
the four valency electrons to form covalent bonds hence do not have delocalized elect
conduct electricity
2. a) Carbon (IV) oxide (CO2) 1
b) 2NaHCO3(s) Na2CO3(s) + H2O(l) + CO2(g) 1
c) – Paper manufacture 1
– Manufacture of glass.
– Softening of hard water.
3. Magnesium has a higher affinity for combined oxygen that carbon./Mg is more reactive
than carbon thus displaces it from its oxide.
4 a) Carbon (iv) Oxide
b) Blue flame. Carbon (iv) oxide burns in air with a blue flame 1
5. a) A brown solid is formed
b) CuO(g) + C(g) ___________ Cu(g) + CO(g)
c) As a fuel in water gas
6. (a) Covalent bond is bond between non-metal atoms where shared electrons are donated equally
by all the atoms involved.
 Dative bond is a bond in which shared electrons are donated by one atom.
Dative bond is a bond in which shared electrons are donated by one atom.
(b) The presence of triple bond in nitrogen requires very high temperatures to break
7. (a) Reduction by using carbon
b) J, carbon and H
decreasing order of reactivity 7. Study the structures A and B:
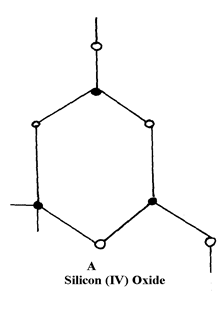
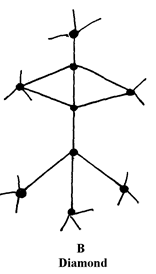
 8. (i) Have giant atomic structure
8. (i) Have giant atomic structure
 (ii) To make drill bits or used in jewellery (any one)
(ii) To make drill bits or used in jewellery (any one)
9. (a) Allotropy is the existence of an element √1 in more than one form without change of state.
(b) Graphite contains delocalized √1 electrons between the layers while diamond has no
3 free √1 electrons. Its atoms are strongly bonded.
10. (a) C(s) + CO2(g) 2CO(g) √1 (1 mk)
(b) Burn charcoal in sufficient √1 oxygen Carbon (II) oxide 3
(being a reducing agent) is easily oxidized to carbon (IV) oxide.√1 (1 mk)
11. (a) Black√ ½ solid changes to reddish brown√ ½
(b) CuO(s) + CO(g) Cu(s) + CO2(g)
√ (1 mk) 2
12. (a) Difference forms of a substance at the same physical state;
(b) In graphite each carbon is bonded to 3 others and there are Vander waals forces between
hexogous;
– In diamond each carbon atom is covalently bonded to four others making a rigid mass;
13. a) – Copper (ii) oxide changes √ ½ from black to brown/ reddish brown/ red brown√ ½
– A white ppt forms in the boiling tube √ ½
b) CO2(g) + Ca(OH)2(aq) _______ CaCO3(g) + H2O(l)
√ 1
c) Unreacted carbon (ii) Oxide is poisonous/ toxic/ pollutant it is converted to the less harmful
gas CO2
14. a) A the substance is a gaining kinetic energy making it to vibrate vigorous up B,
at point B to C the kinetic energy a gained is used to beak down the particle in solid state at
this point the substance start melting and the temperature is constant.
d) It is not water because the melting of water is 1000c not 1150c.
e) The melting point will be lower because of the impurity Nacl.
f) The temperature is constant.
15. (a) (i) Carbon (II) Oxide or CO – (reject Carbon monoxide)
(ii) Combines with haemoglobin to form caborhaemoglobin which prevents carrying of
oxygen
(b) (i) CO(g) + C(s) 2CO(g)
(ii) ZnO(s) + CO(g) Zn(s) + CO2(g)
(c) Orange/yellow Lead (II) Oxides turns grey
(d) CaCO3(s) + 2HCl(aq) CaCl2(aq) + CO2(g) + H2O(l)
(e) Methanoic acid and concentrated sulphuric acid
(f)
16. (a) (i) – Ammonia gas 1
– Calcium carbonate. 1
– Brine 1 or Concentrated sodium chloride.
– Coke (Any three materials)
(ii) – Carbon (IV) oxide. 1
- Ammonia gas. 1
- Water (Any two)
(iii) Chamber 3 1
Chamber 2 1
(iv) U – Ammonia chloride 1
V – Sodium hydrogen carbonate. 1
(b) (i) HN3(g) + H2O(l) + CO2(g) + NaCl(aq) NH4Cl(aq) + NaHCO3(s)
OR
NH3(g) + H2O(l) + CO2(g) NH4HCO3(aq)
NH4HCO3(aq) + NaCl(aq) NH4Cl(aq) + NaHCO3(s)
(ii) NaHCO3 Na2CO3(s) + CO2(g) + H2O(l)
(iii) Ca(OH)2(s) + 2NH4Cl(aq) CaCl2 + 2NH3(g) + 2H2O(l)
c) – Manufacture of glass.
– Softening of hard water.
– Manufacture of papers.
– Manufacture of soap.
– Refining of metals.
17. (a) (i) – The gas is collected over water
– The gas is not passed through a drying agent
 (ii) PbCl2 is formed which is insoluble hence prevents contact between the carbonate and the acid
(ii) PbCl2 is formed which is insoluble hence prevents contact between the carbonate and the acid
(iii) CO2(g) + C(s) 2CO(g)
CO2(g) + 2NaOH(aq) Na2CO3(aq) + H2O(l)
(iv) – Solid CO2 used as a refrigerant
- Used in making aerated drinks
- Solid CO2 is used in cloud-seeding
- CO2 used as an ingredient/air material in solvary process
(v) – Denser than air
– Does not support combustion (burning)
 (b) Reducing Property
(b) Reducing Property
(c)- Al2(CO3)3 hydrolyses in water/moisture forming H+ ions which reacts with the carbonate
 and dissolves
and dissolves
(d) (NH4)2 CO3(s) NH3(g) + CO2(g) + H2O(g)
18. Brown fumes of a gas are produced as the charcoal dissolves in the acid. The charcoal
reduces nitric (V) acid to nitrogen (IV) oxide gas that is brown while the charcoal is oxidized
to carbon (IV) oxide.
19. (a) Due to formation of calcium hydrogen carbonate which is a soluble salt
(b) 2CaCO3(s) + 2CO2(g) + 2H2O 23Ca(HCO3)2(aq)
(- Award 1mk if equation is correctly balanced
– Penalize ½ mk if equation if not balanced)
20. a) A – Concentrated sulphuric acid (vi) acid √1
b)
c) HCOONa(s) + H2SO4 HCOOH (L) + NaHSO4(S)
Hence; HCOOH(l) CO(g) + H2O(L)
Accept conc H2SO4 (reject where concentrated is not mentioned)
Workability √1
Correct method of collection√1
Of the gas √1
The two equations should be mentioned 2 mks





